A crystal ball in the fight against esophageal cancer
May 20, 2022
Researchers from Tokyo Medical and Dental University (TMDU), Beckman Research Institute of City of Hope, Kumamoto University, and Nagoya University develop a liquid biopsy that can effectively determine if a patient with esophageal cancer will respond to certain treatments
Tokyo, Japan – Biomarkers are like the X on a treasure map, but instead of indicating the location of unearthed riches, they indicate an individual’s health status. Common biomarkers include certain RNA or protein molecules present in tissues and body fluids that can show doctors that a patient has a specific disease or may respond well to a particular treatment plan. Liquid biopsies are appealing as a non-invasive method for measuring biomarker levels in a body fluid. In a recent article published in Annals of Surgery, a team led by researchers at Tokyo Medical and Dental University (TMDU) report how they designed such a method for predicting patient outcomes following treatment for esophageal squamous cell carcinoma (ESCC).
ESCC is an aggressive and deadly disease. Data from the United States suggest that its 5-year overall survival rate is less than 20%. Surgery is the typical treatment method, but recently researchers have shown that adding neoadjuvant therapy (NAT) can improve patient survival rates. NAT involves shrinking a tumor using approaches such as chemotherapy or radiation prior to surgery. However, not all patients are responsive to NAT. Unnecessarily performing it can patient time for these patients and lead to worse outcomes. Therefore, the TMDU group aimed to identify biomarkers and characteristics that could preemptively indicate an ESCC patient’s response to NAT.
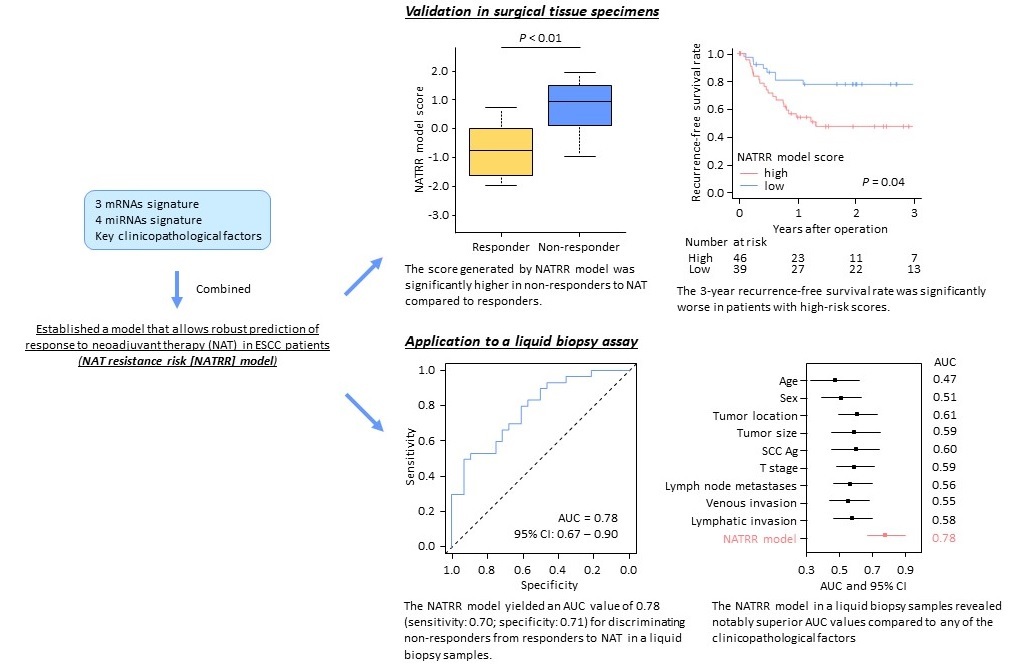
Establishment of a liquid biopsy-based model that allows robust prediction of response to neoadjuvant therapy (NAT) in ESCC patients
NAT resistance risk (NATRR) model were generated using 3 mRNAs signature, 4 miRNAs signature, and key clinocopathological facters, followed by PCR-based validation in surgical tissue specimens. NATRR model were finally applied to a liquid biopsy assay by analyzing pretreatment serum specimens.
“Molecular biomarkers can be extremely useful when making clinical decisions,” says lead author of the study Keisuke Okuno. “We also wanted to translate these biomarkers into an easy-to-use liquid biopsy.” To do this, the team obtained 128 ESCC tissue samples and 58 matched serum samples from two clinical institutions. The serum was obtained prior to any treatment. NAT for these patients involved chemotherapy with 5-fluorouracil and cisplatin, followed by surgery 3 to 5 weeks later. Pathologists then analyzed the surgical specimens for residual tumor cells, which helped them categorize these ESCC patients as completely, partially, or non-responsive to NAT.
“We then examined these patient samples for expression levels of molecules that included three mRNAs and four microRNAs,” states Ajay Goel, senior author of the article. “In previous studies, this panel of mRNAs and microRNAs was found to effectively predict ESCC patient response to NAT.”
After the TMDU group analyzed their data, they concluded that their panel of mRNAs and microRNAs had significant predictive potential. Interestingly, combining the panel with tumor size data further increased the predictive power. mRNA/microRNA signature was the most effective clinical characteristic amongst various combinations. It worked in both early- and advanced-stage ESCC patients.
“We then utilized the pre-treatment serum samples to translate this signature into a liquid biopsy assay, and our approach still maintained its strong ability to predict response to NAT,” explains Okuno.
Overall, the liquid biopsy developed by this team may serve as a non-invasive method for predicting ESCC patient response to NAT. This can lead to precision medicine approaches that will significantly improve the prognosis of many patients who have this disease.
The article, “A transcriptomic liquid biopsy assay for predicting resistance to neoadjuvant therapy in esophageal squamous cell carcinoma,” was published in Annals of Surgery at DOI: 10.1097/SLA.0000000000005473
“We then examined these patient samples for expression levels of molecules that included three mRNAs and four microRNAs,” states Ajay Goel, senior author of the article. “In previous studies, this panel of mRNAs and microRNAs was found to effectively predict ESCC patient response to NAT.”
After the TMDU group analyzed their data, they concluded that their panel of mRNAs and microRNAs had significant predictive potential. Interestingly, combining the panel with tumor size data further increased the predictive power. mRNA/microRNA signature was the most effective clinical characteristic amongst various combinations. It worked in both early- and advanced-stage ESCC patients.
“We then utilized the pre-treatment serum samples to translate this signature into a liquid biopsy assay, and our approach still maintained its strong ability to predict response to NAT,” explains Okuno.
Overall, the liquid biopsy developed by this team may serve as a non-invasive method for predicting ESCC patient response to NAT. This can lead to precision medicine approaches that will significantly improve the prognosis of many patients who have this disease.
###
The article, “A transcriptomic liquid biopsy assay for predicting resistance to neoadjuvant therapy in esophageal squamous cell carcinoma,” was published in Annals of Surgery at DOI: 10.1097/SLA.0000000000005473
Summary
Tokyo Medical and Dental University (TMDU) researchers developed a non-invasive method for predicting the response of patients with esophageal squamous cell carcinoma (ESCC) to tumor shrinkage efforts prior to surgery. Patient response was effectively determined by measuring their serum levels of certain molecules, specifically three mRNAs and four microRNAs, combined with evaluating the tumor size. Use of this novel liquid biopsy could significantly improve the survival rates of patients with ESCC.
Journal Article
JOURNAL:Annals of Surgery
TITLE: A transcriptomic liquid biopsy assay for predicting resistance to neoadjuvant therapy in
esophageal squamous cell carcinoma
DOI:https://doi.org/10.1097/SLA.0000000000005473
TITLE: A transcriptomic liquid biopsy assay for predicting resistance to neoadjuvant therapy in
esophageal squamous cell carcinoma
DOI:https://doi.org/10.1097/SLA.0000000000005473
Correspondence to
Kinugasa Yusuke,MD, PhD, Professor
Department of Gastrointestinal Surgery,
Graduate School of Medical and Dental Sciences,
Tokyo Medical and Dental University (TMDU)
E-mail:kinugasa.srg1(at)tmd.ac.jp
*Please change (at) in e-mail addresses to @ on sending your e-mail to contact personnels.
Department of Gastrointestinal Surgery,
Graduate School of Medical and Dental Sciences,
Tokyo Medical and Dental University (TMDU)
E-mail:kinugasa.srg1(at)tmd.ac.jp
*Please change (at) in e-mail addresses to @ on sending your e-mail to contact personnels.

