“Synthetic polymer-based elucidation of survival strategies of cancer stem cells”
- New insights into the establishment of cancer stem cell-targeted therapy to eradicate cancer by disrupting self-construction of cancer stem cell niche –
Points
| ・ | Cancer stem cell (CSC) niche maintains CSCs, key drivers of cancer progression and relapse. |
| ・ | Polymer array screening identified a CSC niche-mimicking polymer, on which two niche components involved in cell-extracellular matrix (ECM) interaction and iron metabolism were revealed. |
| ・ | ECMs are supplied by the vascular endothelial cells which include CSC-derived ones, while iron is stored in tumor infiltrating macrophages (Mφs) induced potently by CSCs. |
| ・ | These findings demonstrate the utility of synthetic polymers as a tool to establish CSC-targeted therapy for cancer eradication. |
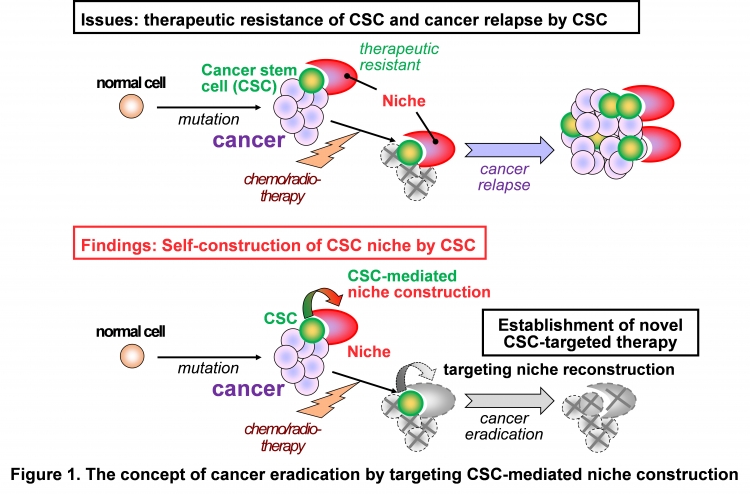
Professor Tetsuya Taga and Assistant Professor Kouichi Tabu at the Department of Stem Cell Regulation, Medical Research Institute, Tokyo Medical and Dental University (TMDU), in collaboration with University of Edinburgh and National Cancer Center Research Institute have identified a niche-mimicking polymer for brain tumor stem cells and revealed the prominent capacities of CSCs to accelerate tumor expansion by self-constructing their own niche (Figure 1).
Background
Cancer stem cells (CSCs) serve as key drivers in cancer progression, recurrence and therapeutic-resistance, and are often proposed as a promising target for cancer eradication. The surrounding microenvironment called niche maintains the CSC pool, where CSCs are retained in their self-renewable and undifferentiated state. Disrupting the CSC niche is theoretically reasonable to impair the self-renewal of CSCs and thereby inhibit the cancer progression and recurrence. However, although being studied by conventional cell and molecular biological approaches, the molecular and cellular basis of CSC niche formation remains to be fully elucidated. The research group has applied synthetic polymers as cell scaffolds to unveil CSC niche and succeeded in identifying a CSC niche-mimicking polymer.
Summary of findings
By using CSCs of glioma, the most common brain tumor, we first screened for niche mimicries that preferentially support the proliferation of CSCs (Figure 2), and succeeded in identifying a urethane-based synthetic polymer PU10. Cells adherent to PU10 exhibited dramatically higher tumorigenic activity when transplanted into immuno-deficient mouse brain, suggesting that PU10 is a CSC niche-mimicking polymer.
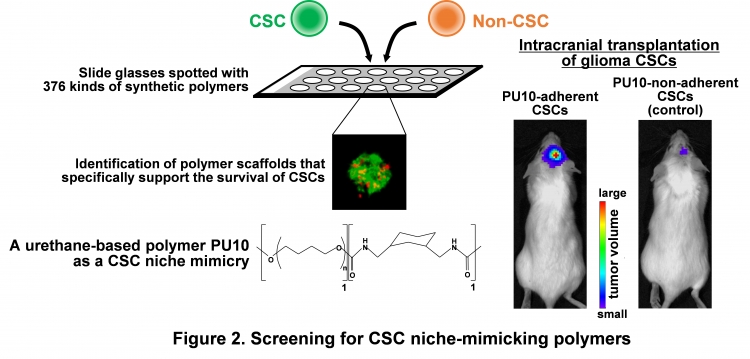
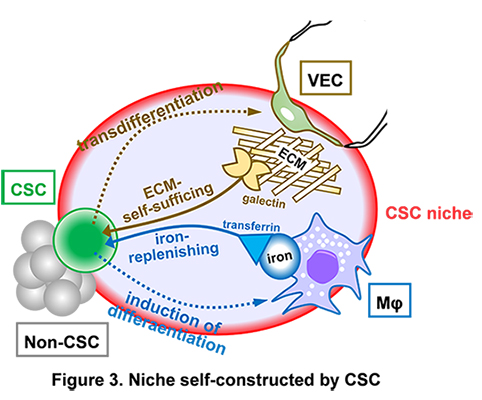
Next, to identify molecular components of CSC niche, proteins captured on PU10 were analyzed, and two molecules, galectin-1 and transferrin which are related to cell-ECM interaction and iron metabolism, respectively, were identified. Examination of tumor tissues generated by intracranial transplantation of CSCs into mice showed that ECMs such as collagen IV and laminin are present around vascular endothelial cells (VECs) and that iron is stored in tumor-infiltrating host macrophages (Mφs).
Clinical data of glioma patients obtained from online database showed that the expression levels of genes related to these niche components, i.e. ECMs, iron and Mφs, are extensively correlated with human glioma malignancies including WHO grading and poor survival.
Clinical data of glioma patients obtained from online database showed that the expression levels of genes related to these niche components, i.e. ECMs, iron and Mφs, are extensively correlated with human glioma malignancies including WHO grading and poor survival.
Furthermore, it was found that CSCs in part transdifferentiate into VECs and also induce iron-storing and protumoral Mφs from bone marrow-derived monocytes (Figure 3), indicating that CSCs strategically expand tumors by self-creating advantageous microenvironments.
Significance of this study
The multidisciplinary study by this group presents the first demonstration of the utility of synthetic polymer scaffolds as a tool for cancer (stem cell) research and reveals a survival strategy of CSCs, in which they self-organize their own niche. As our findings suggest that CSCs can adapt them to the changes of environments, our results not only redefine the current concept of anti-CSC niche therapy but also provide insights into the development of novel CSC-targeted therapies for cancer eradication (Figure 1).
Correspondence to:
Tetsuya Taga, Ph.D., Professor and Kouichi Tabu, Ph.D., Assistant Professor
Department of Stem Cell Regulation, Medical Research Institute,
Tokyo Medical and Dental University (TMDU)
M&D tower 24th Floor, 1-5-45, Yushima, Bunkyo-ku, Tokyo, 113-8510, Japan
E-mail: taga.scr(at)mri.tmd.ac.jp (TT) and k-tabu.scr(at)mri.tmd.ac.jp (KT)
*Please change (at) in the e-mail addresses to @ when sending correspondence.
Department of Stem Cell Regulation, Medical Research Institute,
Tokyo Medical and Dental University (TMDU)
M&D tower 24th Floor, 1-5-45, Yushima, Bunkyo-ku, Tokyo, 113-8510, Japan
E-mail: taga.scr(at)mri.tmd.ac.jp (TT) and k-tabu.scr(at)mri.tmd.ac.jp (KT)
*Please change (at) in the e-mail addresses to @ when sending correspondence.

