Tau and PQBP1: Protein Interaction Induces Inflammation in the Brain
Researchers from Tokyo Medical and Dental University clarify the role of cell receptor PQBP1 in brain inflammation induced by the neurodegenerative disease protein Tau
Tokyo, Japan – Just as a home security system can alert a homeowner to the presence of an intruder, a protein called polyglutamine binding protein-1 (PQBP1) found in brain cells can alert the body to the presence of “intruding” viruses like human immunodeficiency virus (HIV). Now, researchers in Japan have shed new light on the role of PQBP1 in the detection of dysfunctional proteins associated with neurodegenerative disorders.
In a new study published in Nature Communications, researchers from Tokyo Medical and Dental University (TMDU) have revealed the role of intracellular receptor PQBP1 in response to Tau, a protein found primarily in neurons that plays a key role in the progression of neurodegenerative disorders like Alzheimer’s disease.
PQBP1 has been previously shown to sense and bind HIV DNA and trigger an immune pathway known as the cGAS-STING pathway to initiate an inflammatory response. While PQBP1 has also been shown to interact with dysfunctional proteins such as those implicated in the neurodegenerative disorder Huntington’s disease, the specific role of PQBP1 in neurodegenerative inflammatory responses was unclear. To better understand this relationship, researchers from TMDU set out to clarify the nature of the interaction between PQBP1 and Tau.
“By characterizing the relationship between PQBP1 and Tau, we were able to clarify a mechanism of inflammation in the brain that functions in both viral infection and neurodegenerative disease,” says senior author of the study, Hitoshi Okazawa (Figure 1).
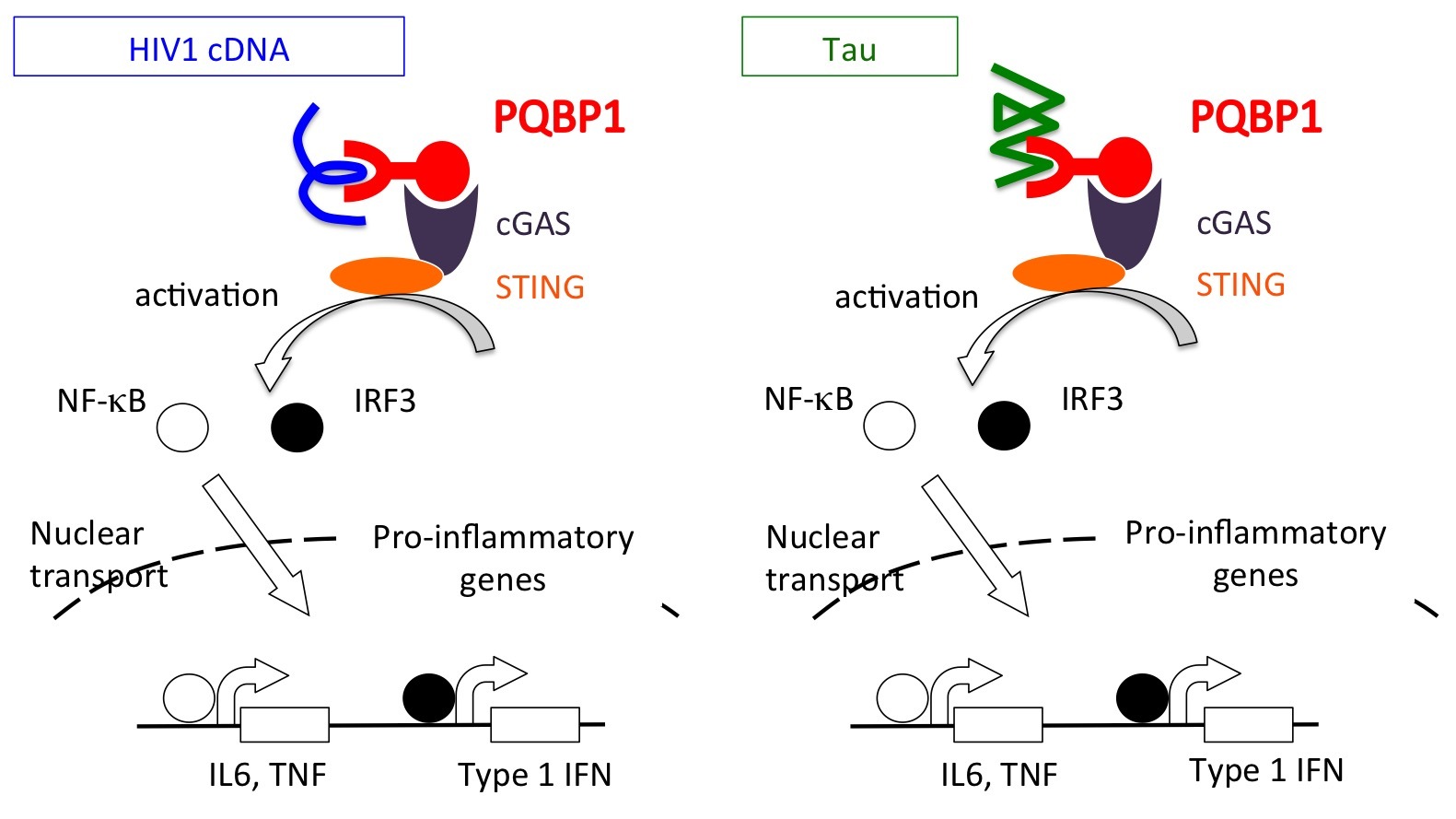
PQBP1, a new intracellular receptor for viral infection and neurodegeneration.
PQBP1 functions in brain microglia, which corresponds to dendritic cells or a part of macrophage regulating innate immune system, as an intracellular receptor for HIV cDNA and Tau proteins similarly.
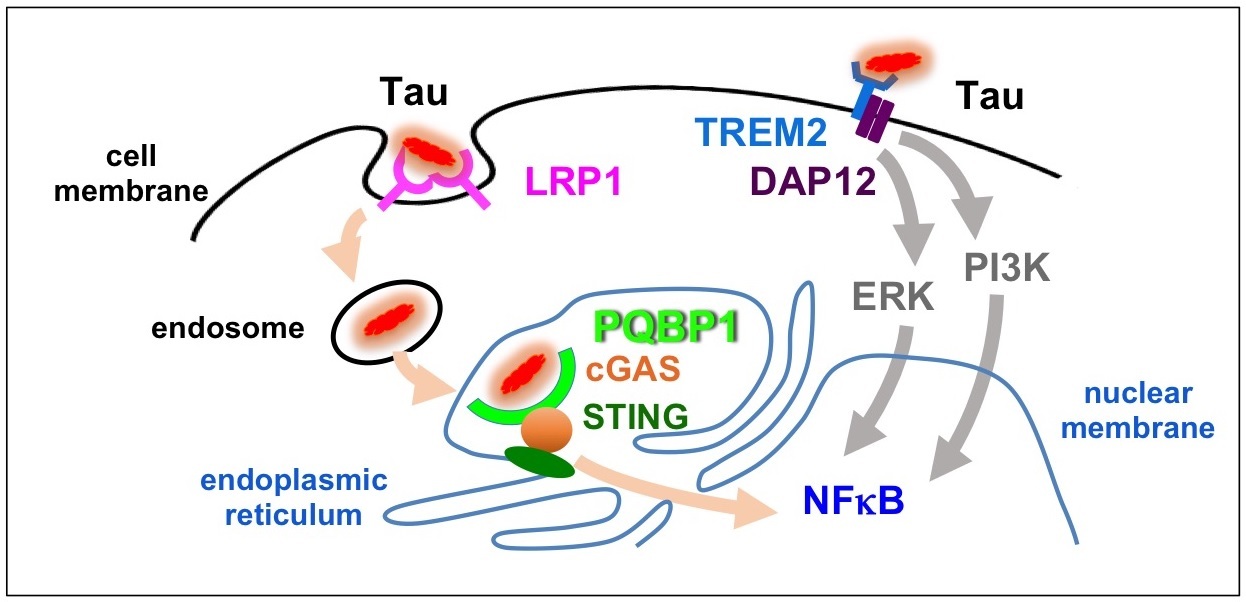
Two pathways independently activate microglia.
Tau protein is incorporated into microglia by LRP1 cell surface receptor and then binds to PQBP1 to activate cGAS-STING pathway and expression of pro-inflammatory genes. Meanwhile, Tau binding to TREM2 activates other signal pathway.
They went on to use a mouse model in which PQBP1 was conditionally inactivated in microglia to show that expression of PQBP1 is necessary for a Tau-induced inflammatory response in vivo.
“We were pleased to find that inactivating PQBP1 in microglia in the mouse model reduced brain inflammation in response to the injection of Tau into the brain,” says Okazawa.
The team further found that mutations in the PQBP1-binding regions of Tau reduced the inflammation in the brain in response to Tau injection. These findings indicate that PQBP1 may represent a potential target for the development of therapeutics for the treatment of Tau-mediated neurodegenerative diseases.
###
The article, “Tau activates microglia via the PQBP1-cGAS-STING pathway to promote brain inflammation,” was published in Nature Communications at DOI: 10.1038/s41467-021-26851-2.
Summary
Journal Article
TITLE:Tau activates microglia via the PQBP1-cGAS-STING pathway to promote brain inflammation
DOI:https://doi.org/10.1038/s41467-021-26851-2

