"Opening Up a Drug Delivery Route – Discovery of a New Vehicle Peptide"
Tokyo – In the field of anticancer therapeutics, cationic antimicrobial peptides (AMPs) have gained prominence because of their cytotoxic and anticancer activity in drug-resistant cancer cells. Peptides are compounds consisting of two or more amino acids linked in a chain.
A membrane impermeable proapoptotic domain (PAD) peptide [KLAKLAK]2 is known to be an antimicrobial agent and to cause mitochondrial membrane disruption, followed by cell death. Most AMPs including PAD have antitumor functions because of a multifunctional host defense system of multicellular organisms; however, they cannot penetrate cell membranes, and alone, they have little cytotoxicity.
Previous studies have shown PAD peptides entering cells by their conjugation with cell-penetrating peptides (CPPs). Particularly, a team of Tokyo Medical and Dental University (TMDU) researchers has shown a cyclic decapeptide, termed peptide 1, mimics the dimerization arm of the EGF receptor (EGFR). EGFR is involved in cellular signal transductions and cancer progression, and is overexpressed in many cancer cells. Hence, binding of peptide 1 to EGFR could potentially retard cancer growth.
In its latest study, the team aimed to verify the selectivity of peptide 1 to EGFR and investigated its efficacy as a vehicle for intracellular delivery to cells. The researchers reported their study in Bioconjugate Chemistry.
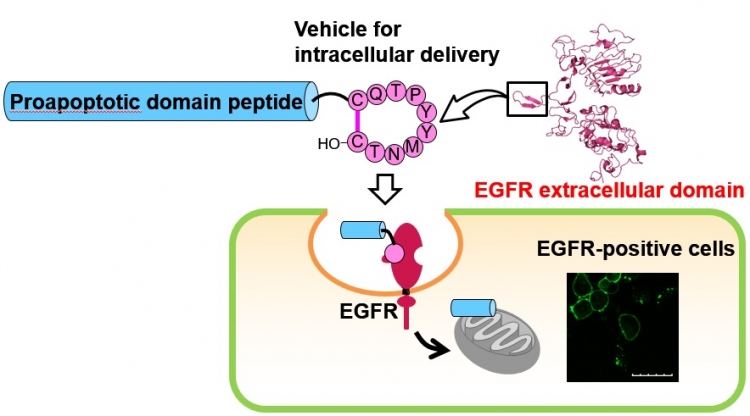
Figure
A cyclic decapeptide, which mimics the dimerization arm of the EGF receptor (EGFR), was previously found to be captured into cells. The authors have found the promising potential of this peptide as an intracellular delivery vehicle directed to EGFR-positive cells. The cellular delivery of the proapoptotic domain (PAD) peptide was challenged by conjugation with the above cyclic peptide. The cellular uptake of the conjugated peptide, which was composed of the cyclic peptide, the PAD peptide and a linker cleavable with a protease, was evaluated by treatment of EGFR-positive cells. Significant suppression of proliferation by the conjugated peptide was shown in a cell viability assay.
Significant suppression of proliferation by peptide 2 was observed in cell viability assays, whereas conjugate 3, which does not contain peptide 1, had no significantly inhibitory activity in the cell lines. Notably, the PAD peptide alone had no effect on the cells.
“Our findings suggest peptide 1 is a promising lead compound as a new intracellular delivery vehicle for therapeutically effective peptides,” senior author Hirokazu Tamamura says. “Also, conjugate 2, with its apoptosis induction activity, can be explored as a new apoptotic peptide that could contribute to the development of anticancer reagents based on the AMP functions.”
The article, “Delivery of a Proapoptotic Peptide to EGFR-Positive Cancer Cells by a Cyclic Peptide Mimicking the Dimerization Arm Structure of EGFR” was published in Bioconjugate Chemistry at DOI:10.1021/acs.bioconjchem.8b00250.
Summary:
Correspondence to:

Department of Medicinal Chemistry,
Institute of Biomaterials and Bioengineering,
Tokyo Medical and Dental University(TMDU)
E-mail:tamamura.mr(at)tmd.ac.jp

