”Signaling Pathway Controls Body Clock and Movement in Adult Mice”
Tokyo, Japan – Signaling pathways involving JNK proteins and their activators MKK7 and MMK4 control several biological processes in response to environmental stimuli, growth factors, and hormones. Previous work has identified particular roles for these pathways in the development of the nervous system, but their effect on the nervous systems of mature animals was unknown. Cell culture studies that inhibited JNK or MKK7 expression increased the lengths of the molecular cycle of gene expression, so-called circadian transcription. However, the function of MKK7–JNK signaling on circadian rhythms in whole animals had not been investigated.
An international partnership between Tokyo Medical and Dental University (TMDU) scientists and colleagues from California, Austria, and other parts of Japan found that MKK7–JNK signaling is essential for control of the mouse circadian clock and the maintenance of nerve function and movement in adult animals.
Because MMK7 and MKK4 regulate all three forms of JNK proteins, removal of either MMK7 or MKK4 expression or that of both proteins can be used to study JNK function. The researchers bred mice in which MKK7 expression was specifically knocked out in nerve cells (neurons) but not the glial cells surrounding and supporting the neurons.
The MMK7 knock-out (KO) mice had greatly enlarged brains compared with control animals because of a larger area of white matter and more junctions between the nerve cells. They also had differences in their behavioral rhythms. “We compared KO mice kept in light–dark conditions to mimic day and night with those kept in constant darkness,” co-author Tokiwa Yamasaki says. “Mice kept in the dark tended to have longer behavioral rhythms which were controlled by their internal circadian clock, while those in light–dark conditions showed typical 24-hour patterns that were regulated by the light.” KO mice were also less active than controls.
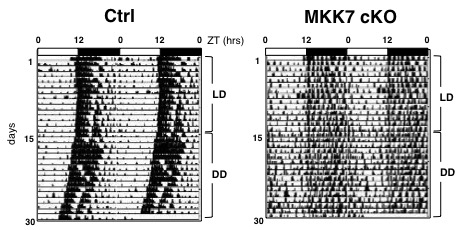
Fig.1 Representative actograms of locomotor activity in MKK7 cKO and control mice at 2.5 to 4 months.
We monitored the free running locomotor activity of MKK7 cKO mice in home cages with infrared area sensors under light and dark (LD) conditions followed by a constant dark (DD) condition. In the DD condition, activity becomes regulated by their internal circadian clock. MKK7 cKO mice displayed slightly longer periods of circadian behavioral rhythms than control mice. In addition, the average activity counts under LD conditions were significantly reduced in MKK7 cKO mice compared to control mice.
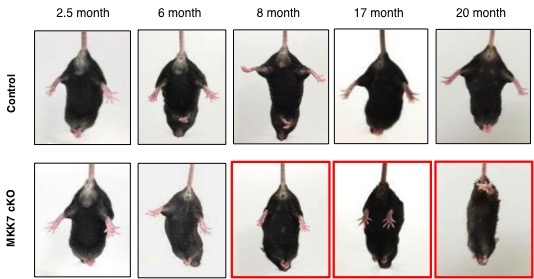
Fig.2 Age-dependent muscle weakness in MKK7 cKO mice.
We analyzed the hind-limb reflex under tail suspension. The angle of the hind-limb was indistinguishable between control and MKK7 cKO mice until 6 month-old. At 8 months, the hind-limb reflex gradually decreased in MKK7 cKO mice. MKK7 cKO mice finally became unable to extend their hind-limb at 20 months of age.
These changes in circadian rhythms and behavioral activity are also seen in some human psychiatric diseases, suggesting that restoring normal MKK7–JNK signaling could help treat such disorders.
The article “Age-dependent motor dysfunction due to neuron-specific disruption of stress-activated protein kinase MKK7” will be published shortly.
Summary Text:
Correspondence to:
Department of Developmental and Regenerative Biology,
Medical Research Institute,
Tokyo Medical and Dental University (TMDU)
Phone:+81-3-5803-4659 Fax:+81-3-5803-0272
E-mail: nishina.dbio (at) mri.tmd.ac.jp

