“Discovery of a novel source of dendritic cells, the control tower of the immune system”

Toshiaki OHTEKI
Professor, Department of Biodefense Research,
Medical Research Institute (right)
Nobuyuki ONAI
Junior Associate Professor,
Department of Biodefense Research,
Medical Research Institute (left)
Highlights
| 1. | Dendritic cells (DCs) maintain immune tolerance under steady-state conditions, and activate immune cells upon infection. |
| 2. | Our research group discovered a progenitor cell type that is strictly committed to the DC lineage. |
| 3. | Each of these progenitor cells can give rise to 500~1,000 DCs. |
| 4. | Our findings may lead to progenitor-based therapeutic applications for viral infections and autoimmune diseases. |
Main findings and significance of this study
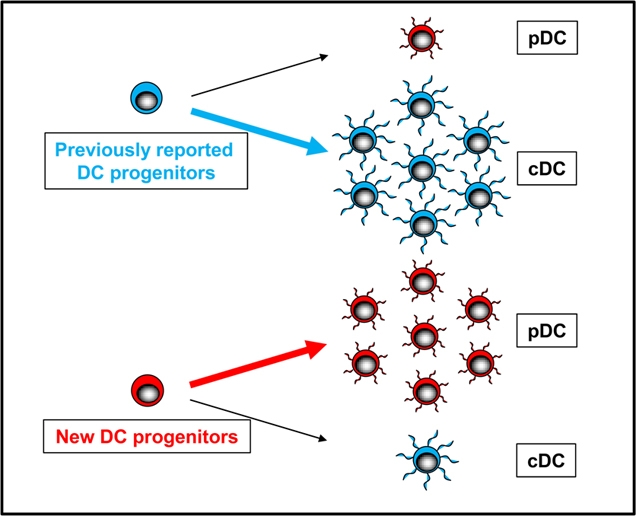
Figure 1. Identification of new DC progenitors.
The new DC progenitors possess a prominent plasmacytoid DC differentiation potential compared with the previously reported DC progenitors.
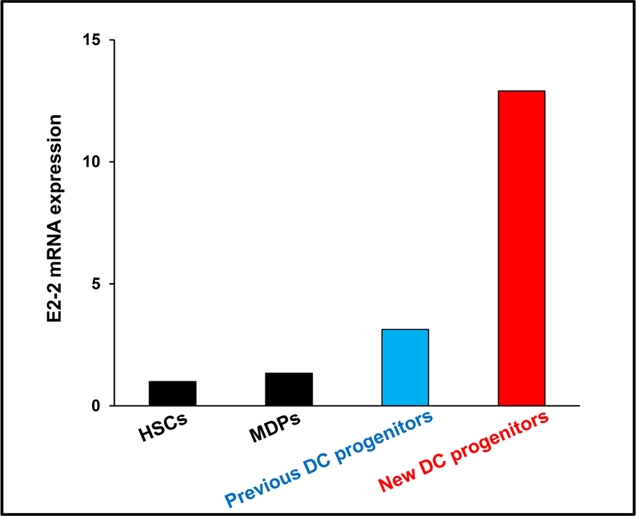
Figure 2. New DC progenitors highly express E2-2.
E2-2 is an essential transcription factor for pDC development. Compared with hematopoietic stem cells (HSCs), macrophage and DC progenitors (MDPs), and the previously described DC progenitors, the new DC progenitors express higher amounts of E2-2.
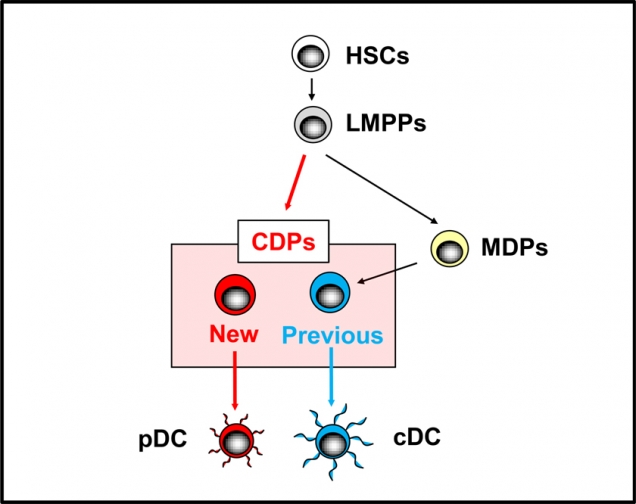
Figure 3. CDPs include the previous and new DC progenitors.
In addition to the previously identified pathway (MDPs to previous DC progenitors), the CDPs appear to be directly derived from lymphoid-primed multi-potent progenitors (LMPPs).
Correspondence to:
Department of Biodefense Research
Medical Research Institute, Tokyo Medical and Dental University
1-5-45 Yushima, Bunkyo-ku, Tokyo 113-8510, Japan
E-mail: ohteki.bre(at)mri.tmd.ac.jp
*Please change (at) in the e-mail addresses to @ when sending correspondence.

