Starvation induced autophagy enhances accumulation of amyloid-beta (Aβ) in neurons (2015)
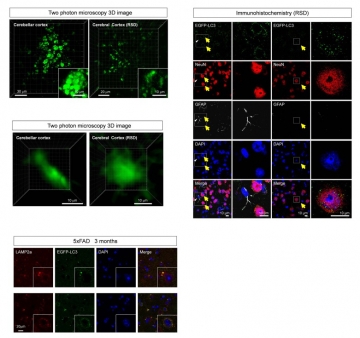
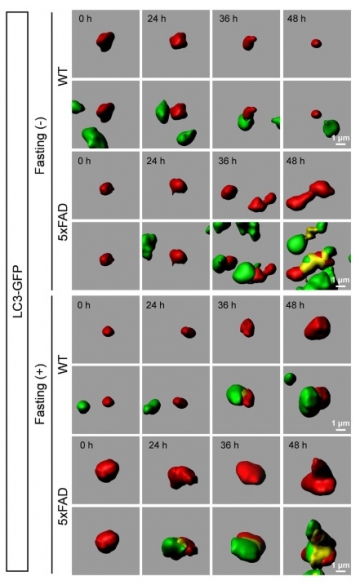
We developed a new technique to observe macroautophagy in the brain in vivo, and examined whether fasting induced macroautophagy in neurons and how the induction was different between Alzheimer's disease (AD) model and control mice. Lentivirus for EGFP-LC3 injected into the brain successfully visualized autophagosome in living neurons by two-photon microscopy. The time-lapse imaging revealed that fasting increased the number, size and signal intensity of autophagosome in neurons. In AD model mice, these parameters of autophagosome were higher at the basal levels before starvation, and increased more rapidly by fasting than in control mice. However, metabolism of exogenous labeled Aβ evaluated by the new technique suggested that the activated macroautophagy was insufficient to degrade the intracellular Aβ increased by enhanced uptake from extracellular space after fasting. Ordinary immunohistochemistry also revealed that fasting increased intracellular accumulation of endogenous Aβ, triggered cell dysfunction but did not mostly decrease extracellular Aβ accumulation. Moreover, we unexpectedly discovered a circadian rhythm of basal level of macroautophagy. These results revealed new aspects of neuronal autophagy in mormal/AD states and indicated usefulness of our method for evaluating autophagy functions in vivo.
Neurodegenerative diseases, including AD, have a unique pathological feature which is abnormal protein accumulation inside or outside of cells. Two distinct abnormal proteins accumulate in AD brain. The senile plaques consist of abnormal protein called beta-amyloid (Aβ) outside cells, and the neurofibrillary tangles that tau protein aggregates inside the cell. On the other hand, two distinct protein degradation systems are well-studied as cellular mechanism to remove abnormal proteins, ubiquitin-proteasome and autophagy systems. Autophagy system is further classified to "basal autophagy" which works at a constant level and "incuded autophagy (macroautophagy)" which is activated in calorie restriction.
Macroautophagy has been known to play a significant role in tissues other than the brain. While areport suggested that macroautophagy in brain tissue is not observed (Mizushima et al, Mol Biol Cell 2004; while there are, etc.) , except for the aggregation of the abnormal protein-induced autophagy by calorie restriction, etc. in neurodegenerative disease, a result of improving the symptoms has been reported by many (Ravikumar et al, Nat Genet 2004; etc.) that the presence or absence of induced autophagy in nerve cells was not settled. Furthermore, our proof of the induced autophagy in higher animals was important, since the signaling pathway from the insulin receptor via mTOR (mammalian target of rapamycin) is considered important, and diabetes and high calorie are said risk factors of Alzheimer's disease pathology. Therefore, in this study it was the first object of the present invention is to clarify the "presence or absence of induced autophagy in nerve cells".

