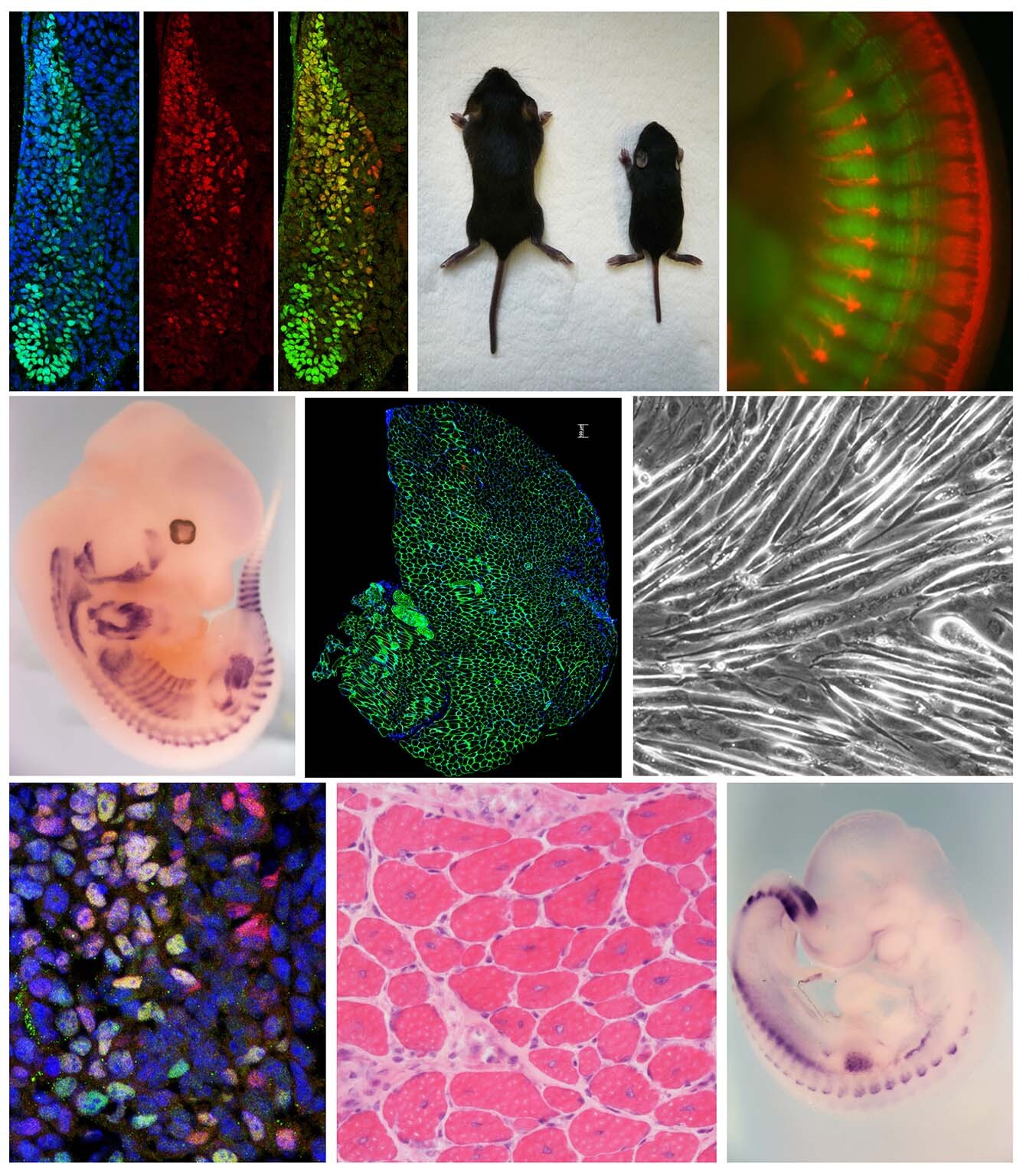
Research
- 1. Elucidation of the Mechanisms Underlying Chronic Inflammation
- 2. Molecular Mechanisms of Age-Related Impairment of Muscle Regeneration and the Development of Therapeutic Strategies for Sarcopenia
- 3. Development of Organoid Technologies and Their Application to the Treatment and Prevention of Age-Related Diseases
We aim to elucidate the etiology and pathophysiology of age-related diseases, including lifestyle-related disorders such as obesity and diabetes, as well as sarcopenia.
Lifestyle-related diseases—including myocardial infarction and cerebral infarction caused by atherosclerosis, as well as diabetes and chronic kidney disease—are increasing worldwide. In parallel, age-related loss of skeletal muscle mass, known as sarcopenia, has emerged as a major factor contributing to a decline in quality of life (QOL) in the elderly.
Our research focuses on understanding the mechanisms underlying these common and clinically important conditions, with the ultimate goal of developing effective novel strategies for their treatment and prevention.
References
1. Elucidation of the Mechanisms Underlying Chronic Inflammation
Chronic inflammation has emerged as a common pathological feature shared by obesity, lifestyle-related diseases, and cancer. It can be regarded as a condition in which inflammation, initially triggered as a protective response to various stresses, fails to resolve properly and instead persists over time. However, the mechanisms by which inflammation becomes chronic remain incompletely understood.
To address this question, we focus on macrophages, which play a central role in the development of chronic inflammation. By integrating molecular biological approaches with cutting-edge transcriptomic and epigenomic analyses, we aim to elucidate the mechanisms that drive the persistence of inflammatory responses. In particular, we are interested in the tight coupling between immune responses and cellular metabolism in macrophages, and we seek to develop novel therapeutic and preventive strategies targeting macrophage metabolism in chronic inflammatory conditions.
More recently, chronic inflammation has been implicated not only in lifestyle-related diseases but also in a broader range of conditions, including chronic endometritis associated with infertility, noise-induced hearing loss, and impaired wound healing. In collaboration with clinicians across multiple specialties, we are working to elucidate the underlying mechanisms of these conditions and to develop innovative therapeutic approaches.
2. Molecular Mechanisms of Age-Related Impairment of Muscle Regeneration and the Development of Therapeutic Strategies for Sarcopenia
Skeletal muscle plays essential roles not only in locomotion and posture maintenance but also as the largest metabolic organ responsible for systemic glucose homeostasis. Muscle atrophy caused by physical inactivity, prolonged immobilization, or aging leads not only to a decline in physical function but also to an increased risk of lifestyle-related diseases, ultimately exerting a profound impact on quality of life. Age-associated loss of muscle mass and strength, termed sarcopenia, has become an increasingly serious health issue in aging societies such as Japan.
In contrast, skeletal muscle possesses a remarkable capacity for regeneration. Following muscle injury, muscle stem cells—known as satellite cells—play a central role in the regenerative process. These cells normally reside in a quiescent state but become activated upon injury, proliferate and differentiate, and ultimately give rise to new muscle fibers. In addition, satellite cells self-renew to maintain their population, thereby preserving tissue homeostasis and regenerative potential.
Muscle injury is also accompanied by a robust inflammatory response, during which various immune cells accumulate in the damaged tissue. Notably, depletion of macrophages severely impairs muscle regeneration, highlighting their critical role in the repair process.
Based on this background, we focus on the interactions between immune cells—including macrophages—and satellite cells. Using cutting-edge approaches such as single-cell analysis, we aim to elucidate the coordinated regulatory mechanisms that integrate inflammation and regeneration following muscle injury. Ultimately, our goal is to establish a foundation for the development of preventive and therapeutic strategies for sarcopenia.
3. Development of Organoid Technologies and Their Application to the Treatment and Prevention of Age-Related Diseases
Pluripotent stem cells, including human induced pluripotent stem (iPS) cells, possess a high capacity for differentiation and can give rise to a wide variety of cell types. In contrast, tissue stem cells play essential roles in maintaining tissue homeostasis by replenishing and repairing cells within specific organs. In recent years, there has been growing interest in the generation of organoids—miniaturized and functional organ-like structures—using these stem cells in vitro.
Organoids are characterized by their ability to self-organize within culture systems. In particular, the organoids we generate are composed of multiple cellular lineages, enabling them to recapitulate key structural features and diverse cellular functions of in vivo organs. We have successfully established multi-organ digestive system organoids that recapitulate developmental processes, including liver formation, as well as skeletal muscle organoids that model tissue regeneration.
These organoids provide powerful platforms for analyzing biological phenomena that are difficult to observe in vivo, and they hold great potential for applications in regenerative medicine and drug screening. We believe that further refinement of organoid technologies will significantly expand their applicability. Accordingly, we are developing novel methodologies for organoid generation and exploring their applications, particularly in the development of therapeutic and preventive strategies for age-related diseases.