Research
- The Hippo pathway and transcriptional coactivators, YAP1 and TAZ.
- The Hippo pathway-dependent and –independent aspects of RASSF proteins.
- The Hippo pathway and RASSF proteins in human cancers.
- Versatile roles of YAP1 and TAZ beyond cancer.
- Chemical and biological approaches to dissect the regulation of YAP1 and TAZ and the functions of C-RASSF proteins.
We have studied the transcriptional coactivators, YAP1 and TAZ (also called WWTR1), which are regulated by the tumor suppressive Hippo pathway, and RASSF proteins, which are closely related to the Hippo pathway. For the details, please refer to the attached references.
We have also started to develop the model mice for the study of ageing. We plan to use these mice to clarify the moleculer mechanism underlying ageing-assoicated muscle atrophy (sarcopenia).
References
- What is the Hippo pathway?
- Mammalian Hippo pathway.
- RASSF6
- Drug targets
The Hippo pathway and transcriptional coactivators, YAP1 and TAZ.
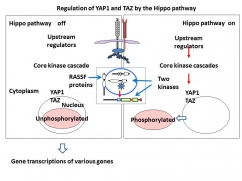
When cells reach confluence or when cells are exposed to DNA damage, the Hippo pathway is activated to phosphorylate YAP1 and TAZ and shut off YAP1- and TAZ-dependent gene transcriptions. Recent studies have revealed that YAP1 and TAZ are regulated by not only the Hippo pathway but also the cell junction proteins, the actin cytoskeleton , and the mechanical stretch (not shown).
The Hippo pathway-dependent and –independent aspects of RASSF proteins.
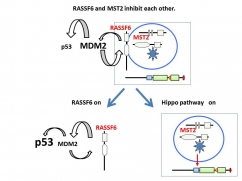
RASSF6 and MST2 form the complex and inhibit each other under the basal condition and MDM2 suppresses p53 expression (The upper part).
RASSF6 is released from MST2 upon the activation of the Hippo pathway and enhances self-ubiquitination of MDM2 to up-regulate p53 (The lower part).
RASSF6 also induces apoptosis through p53-independent mechanism (not shown).
The Hippo pathway and RASSF proteins in human cancers.
Versatile roles of YAP1 and TAZ beyond cancer.

YAP1 and TAZ interact with various transcriptional factors and play versatile roles in physiological and pathophysiological conditions.
Chemical and biological approaches to dissect the regulation of YAP1 and TAZ and the functions of C-RASSF proteins.

Examples of the cell-based assays to identify chemical compounds that activate and inhibit YAP1 and TAZ directly or through the Hippo pathway.
The candidate compounds are further assayed by conventional reporter assays and various phenotypic assays such as myogenesis, adipogenesis, cancer cell motility etc.
To gain further insights, we adopt chemical and biological approaches. We generated various cell-based assays to screen for inhibitors and activators of YAP1 and TAZ. We have obtained several candidate compounds. Some of them regulate YAP1 and TAZ through the Hippo pathway, whereas others work independently of the Hippo pathway. The identification of the molecular targets of these compounds may bring new insights into the regulatory mechanism of YAP1 and TAZ. Needless to say, these candidates provide us with the possibilities to obtain the compounds that are useful to treat various diseases. We are selecting the inhibitors of YAP1 and TAZ that suppress malignant properties of cancer cells in vitro and plan to test them using in vivo transplantation models. We have successfully obtained one TAZ activator that facilitates myogenesis and prevents muscle atrophy. We expect some YAP1 activators to facilitate tissue repairs. The study on C-RASSF functions is more challenging and we still need the basic study to characterize each C-RASSF. Even so we have started to build new methods to monitor C-RASSF expression and plan to discover the compounds that enhance it.

