
Institute of Integrated Research, Institute of Science Tokyo
Department of Advanced Nanomedical Engineering,
Medical Research Laboratory

Research Video
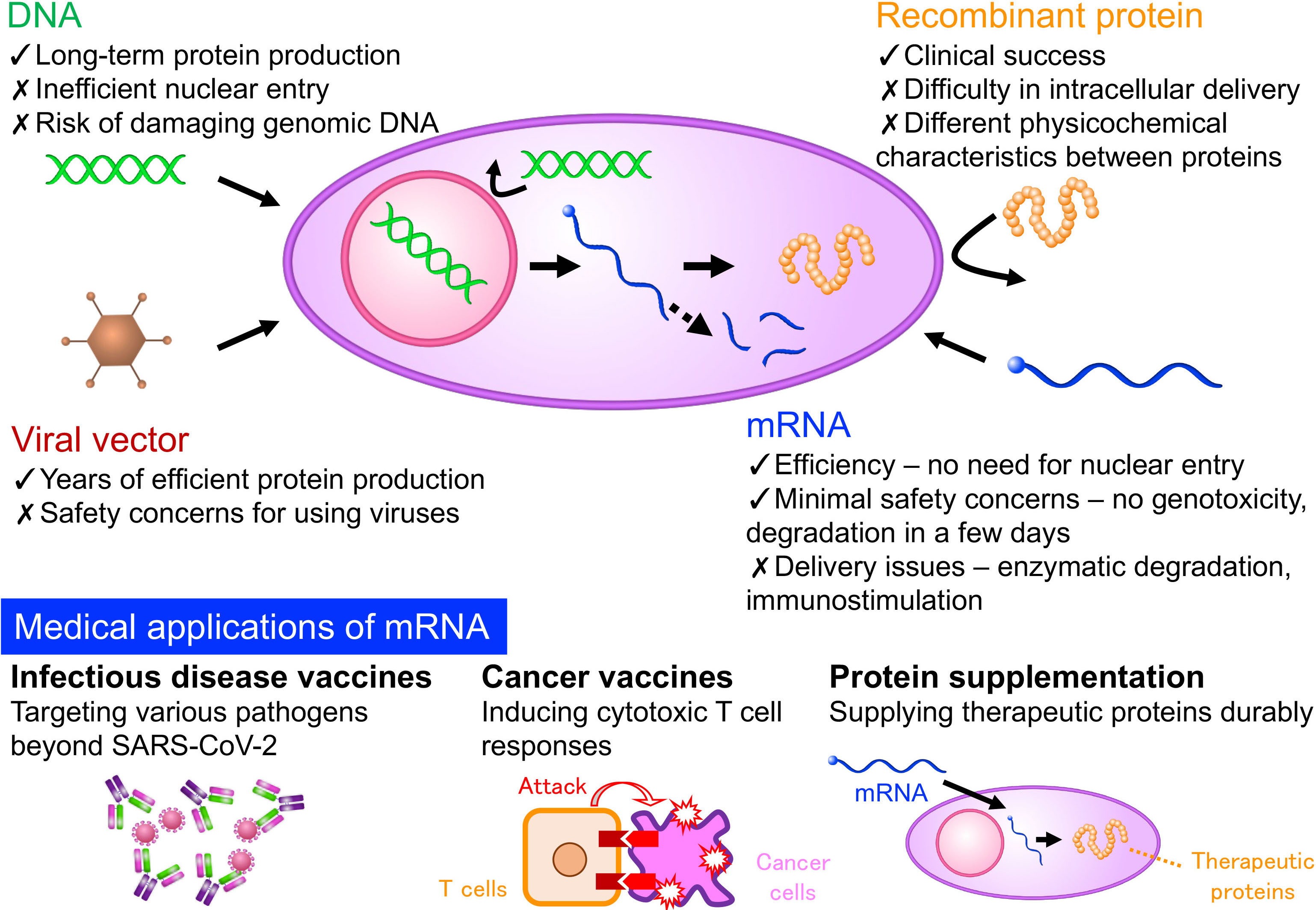
Clinical approvals of nucleic acid therapeutics and gene therapy have increased since the middle of 2010s. mRNA vaccines play a considerable role in suppressing the COVID-19 pandemic. These new drug classes easily apply to numerous diseases by changing the nucleic acid sequences. This advantage prompts further approvals of these drug classes shortly. Among the drug classes, mRNA has several advantages in producing therapeutic proteins in the body. Thus, its application expands beyond infectious disease vaccines to treating numerous diseases. Meanwhile, drug delivery systems (DDS) are essential for mRNA to function appropriately in the patient’s body. In this research field, we develop nano DDS, apply it to disease treatment, and undertake translational research.
Reference: Molecular Pharmaceutics 2020,
Advanced Drug Delivery Reviews 2023
1) Development of nano DDS
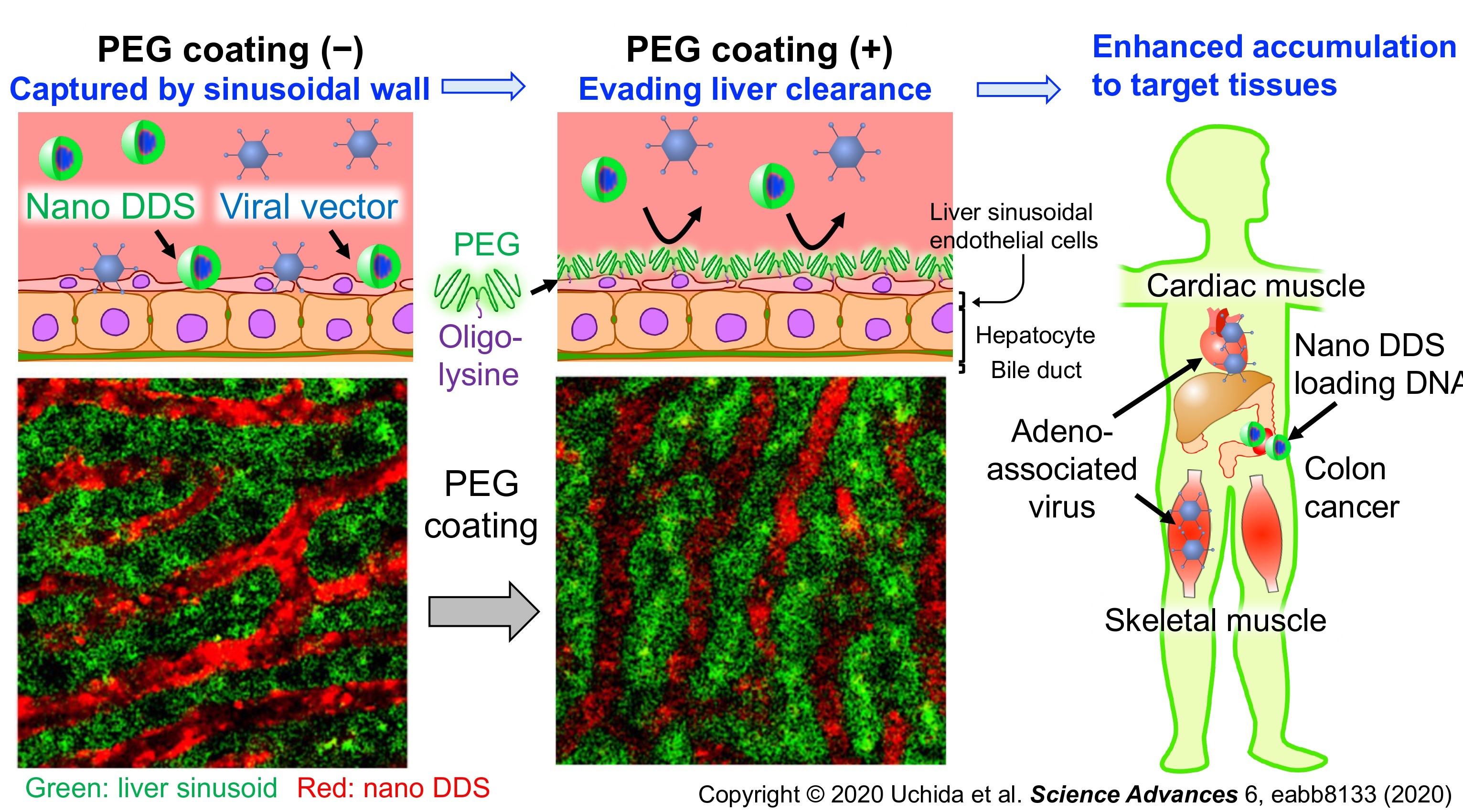
Protecting nanomedicines from adsorption onto liver sinusoidal walls
Even stealth nanomedicines cannot wholly escape from the body’s systems that recognize and clear foreign nanoparticles. Especially, liver sinusoidal walls capture circulating nanomedicines efficiently. Transient poly(ethylene glycol) (PEG)-coating of the sinusoidal walls provides a promising solution. Indeed, this strategy suppressed the sinusoidal clearance of gene therapeutics, including viral vectors and nano DDS loading DNA, and improved their delivery efficiency to the target organs. Now, we are applying this strategy to disease treatment.
Reference: Science Advances 2020
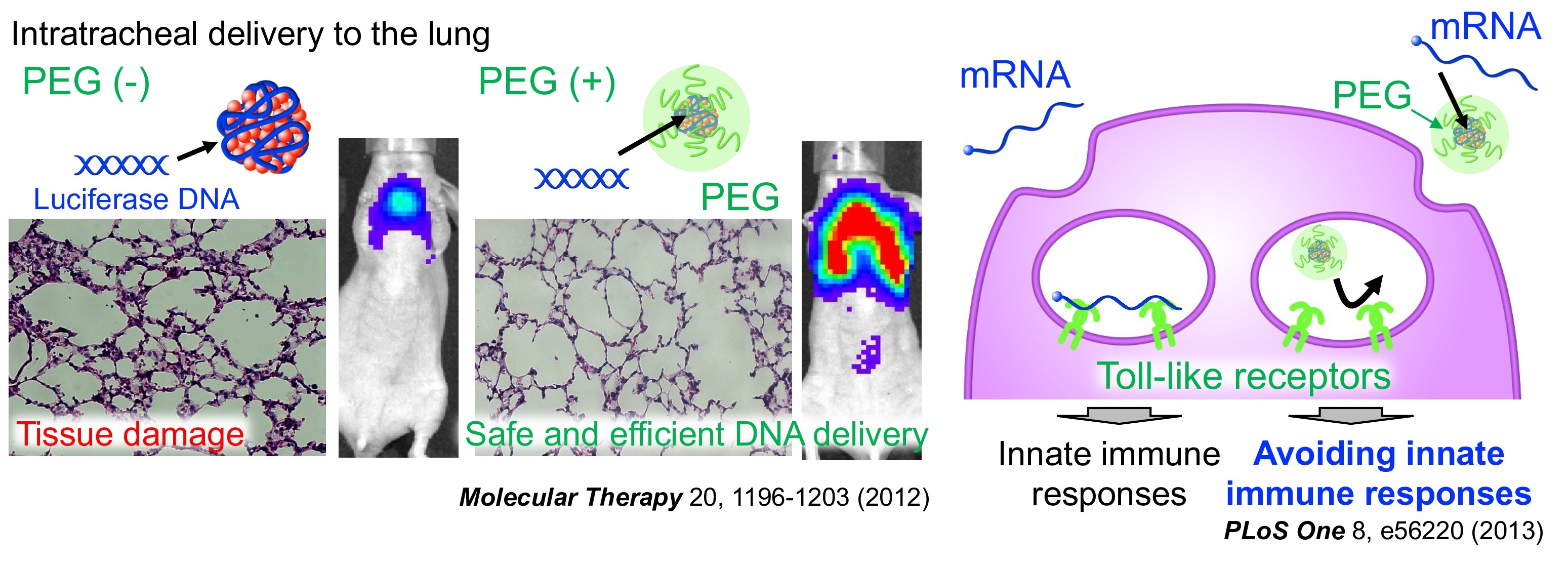
Delivering nucleic acid therapeutics without damaging tissues
Tissue damage and inflammatory responses after nucleic acid delivery can cause adverse effects and hamper the therapeutic process. PEGylation of polymeric nano DDS provides a solution to this issue. PEGylation improves in vivo DNA delivery efficiency with minimal tissue damage. Moreover, in mRNA delivery, PEGylated nano DDS inhibits innate immune receptor recognition of mRNA, allowing mRNA delivery without inflammatory responses. These safety advantages of this system benefit its therapeutic application to tissue protection, repair, and regeneration, as shown in 3) Therapeutic applications.
Reference: Molecular Therapy 2012, PLoS ONE 2013
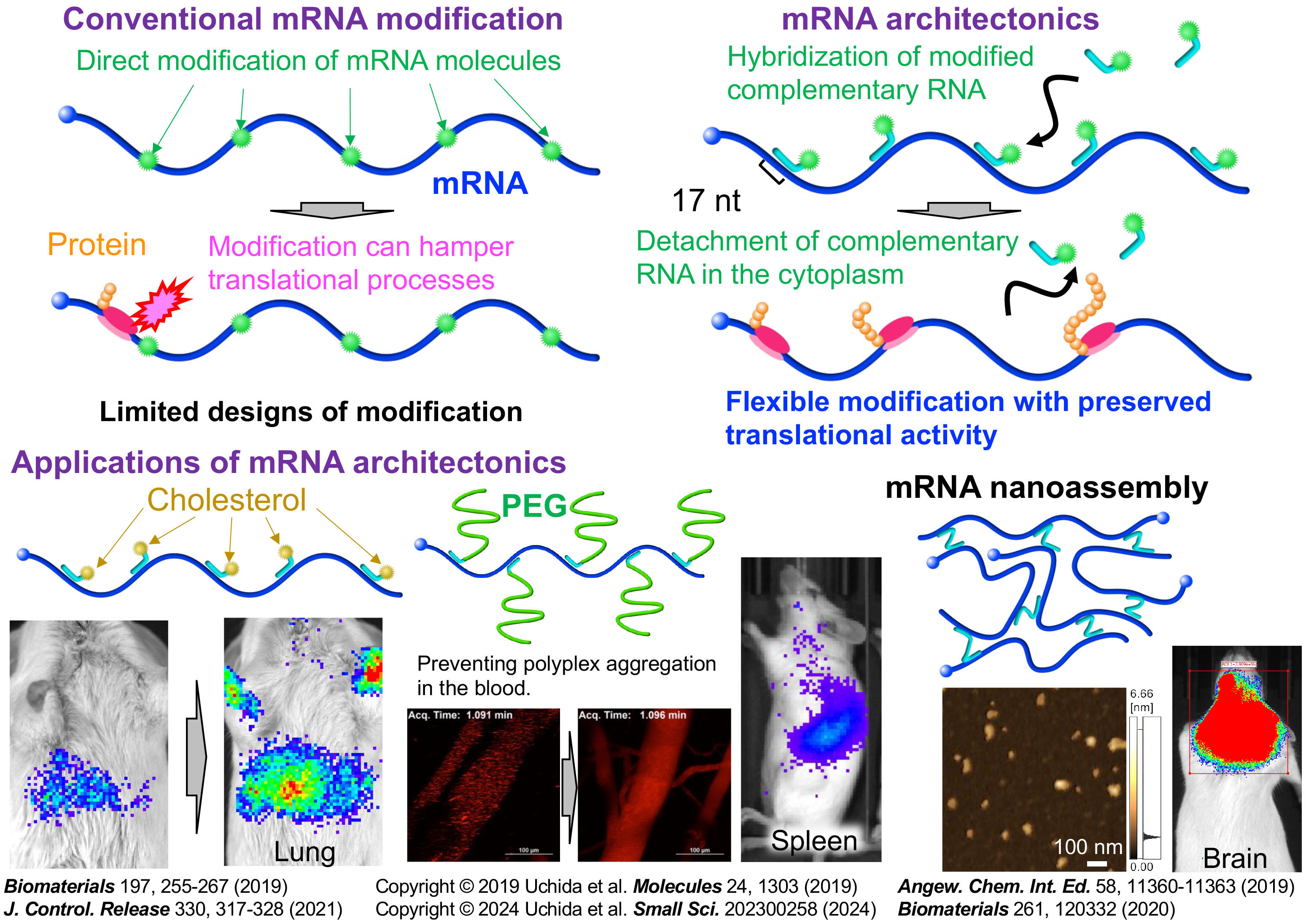
2) mRNA engineering – mRNA architectonics
Most research on mRNA DDS development focuses on designing lipids and polymers rather than mRNA, as mRNA tolerates only modest modification to preserve its translational activity. Instead of directly modifying mRNA molecules, we hybridize functionalized complementary RNA oligonucleotides to mRNA. This mRNA engineering strategy allows various modifications without impairing translational efficiency, improving nano DDS functionalities. Furthermore, we applied this approach to mRNA vaccines by incorporating immunostimulatory adjuvant functionalities to mRNA, as shown in 3) Therapeutic applications.
Reference: Biomaterials 2019, Angewandte Chemie International Edition 2019, Biomaterials 2020
3) Therapeutic applications
mRNA vaccines
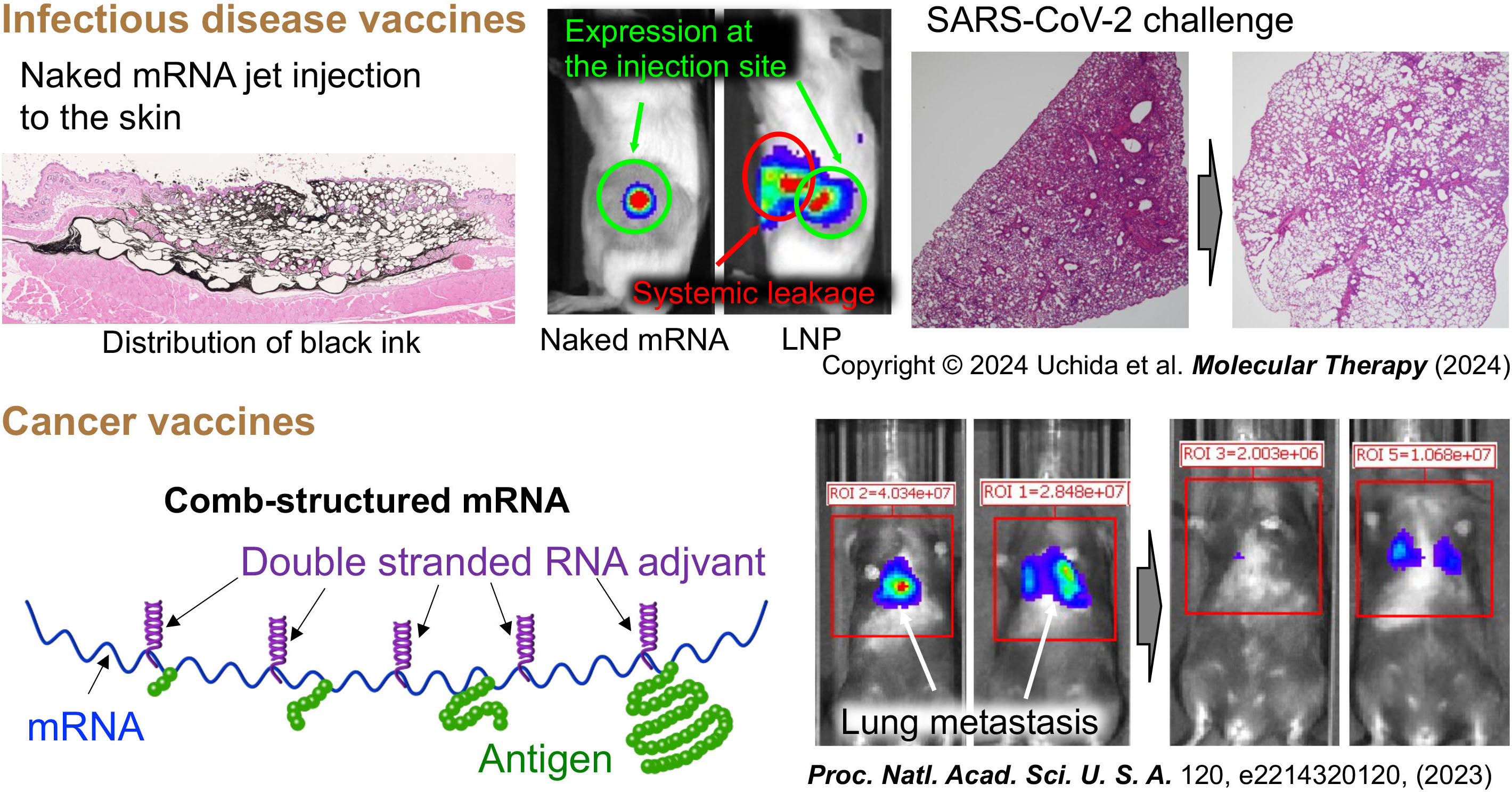
Current mRNA vaccines against infectious diseases raise safety concerns due to the use of delivery carriers. To address this issue, we developed carrier-free mRNA vaccines utilizing jet-injection of naked mRNA. This approach has demonstrated efficacy in protecting mice from SARS-CoV2 infection and eliciting high antibody titers in non-human primates. mRNA vaccines also hold promises for generating immune cells, killing cancer cells. However, the efficacy of mRNA cancer vaccines needs improvement. For this purpose, we incorporated immunostimulatory double-stranded RNA structure into mRNA to prepare comb-structured mRNA. Comb-structured mRNA has dual functionalities of antigen expression and adjuvants, improving the efficiency of cancer vaccines.
Reference: PNAS 2023, Molecular Therapy 2024
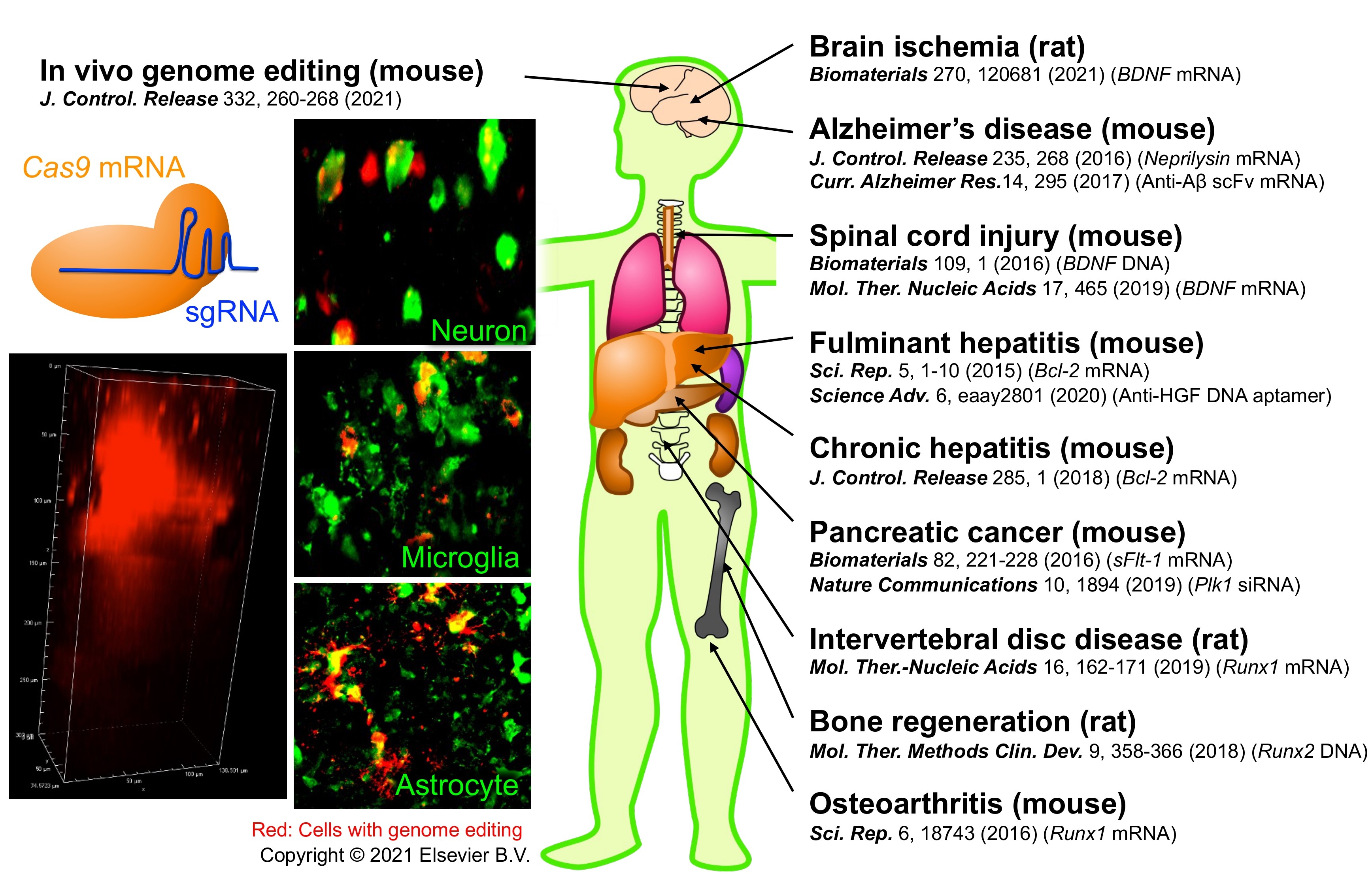
Disease treatment, genome editing
We employ several new drug classes for disease treatment, including mRNA, plasmid DNA (pDNA), DNA aptamer, and siRNA. We also succeed in RNA-based in vivo genome editing in mouse brains. Successful therapeutic applications require a comprehensive overview of drug classes, DDS, and medical needs.
Reference: Biomaterials 2016, Science Advances 2020, Journal of Controlled Release 2021
4) A start-up company
Start-up companies largely contributed to developing COVID-19 vaccines. Prof. Uchida co-founded Crafton Biotechnology, a start-up company, in March 2022 to develop mRNA therapeutics. Researchers from various fields, including nucleic acid chemistry, synthetic biology, data science, medical engineering, and clinical medicine, participate in the company.
Reference: Website