研究業績
原著論文・総説・著書
2026
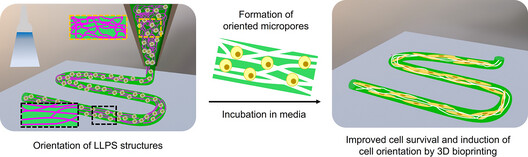 Oriented Micropore-Forming Bioinks for 3D Bioprinting of Muscle Tissues
Oriented Micropore-Forming Bioinks for 3D Bioprinting of Muscle Tissues
Palai, D.; Yasue, H.; Ohta, M.; Uto, K.; Taguchi, T.; Nishiguchi, A.
Small 2026, 22(6), e09439.
Abstract3D bioprinting provides a wide avenue for designing complex and customized constructs for regenerative medicine. Bioink formulations in 3D bioprinting usually lack micrometer-sized and interconnected pores for the supply of nutrients and oxygen and biological communications with host tissues, thus limiting cellular activities and therapeutic efficacy. Herein, we present microfibrous pore-forming bioinks for fabricating microporous hydrogels that encapsulate cells for muscle tissue reconstruction. Using phase separation technology, a liquid porogen is embedded into gelatin-based bioinks to form microfibrous structures. Printing bioinks with shear stress enabled the orientation of microfibrous pores along the printing direction, which facilitated the orientation of printed cells and enhanced myoblast differentiation. Moreover, the porous 3D scaffold exhibited promising results in terms of supplying nutrients and oxygen to improve cell survival. Printed tissue constructs are successfully transplanted into muscle tissue defects. This approach holds immense potential for creating anisotropically oriented 3D tissue constructs for applications in cell transplantation, drug screening, and disease modelling.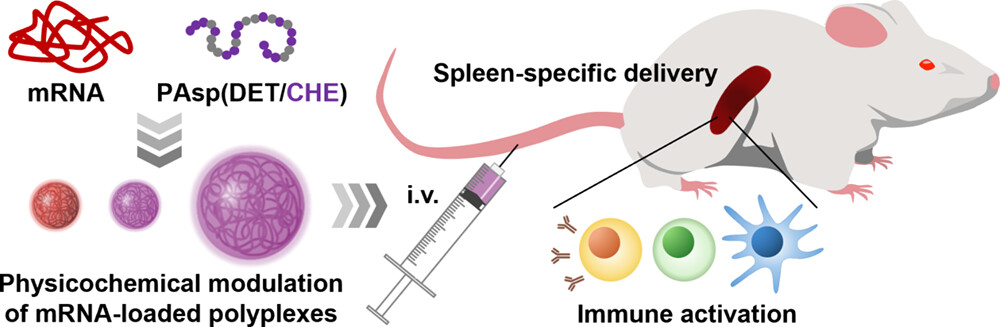 Formulation-Driven Control of mRNA Polyplex Physicochemical Properties Enables Spleen-Targeted Systemic Delivery
Formulation-Driven Control of mRNA Polyplex Physicochemical Properties Enables Spleen-Targeted Systemic Delivery
Hori, M.; Qiao, N.; Yamada, K.; Yum, J.; Ogura, S.; Uchida, S.; Kim, H. J.; Naito, M.; Miyata, K.
ACS Appl. Bio Mater 2026, 9(8), 3878-3886.
Increasing polycation hydrophobicity in mRNA polyplex vaccines enhances the efficacy of humoral and cellular immunity induction
Qiao, N.; Hori, M.; Naito, M.; Chang, H.; Ogura, S.; Suzuki, M.; Kimura, T.; Fukushima, S.; Kim, H. J.; Uchida, S.; Miyata, K.
Biomaterials 2026, 324, 123515.
AbstractPolycation-based mRNA delivery systems, known as polyplexes, hold significant potential for use in mRNA vaccines and therapeutics. However, their performance has been suboptimal in infectious disease vaccines, which require the induction of both humoral and cellular immunity. Herein, we optimized polycation hydrophobicity to maximize the efficacy of humoral and cellular immunity induction, using biodegradable amphiphilic polyaspartamide derivatives as a platform. The side chains of the polymers contain cationic diethylenetriamine (DET) and hydrophobic 2-cyclohexylethyl (CHE) moieties at varying ratios. Increasing the CHE introduction ratio enhanced immunostimulatory adjuvanticity by activating NLRP3 inflammasome pathway, leading to more efficient activation of cultured dendritic cells. Following subcutaneous injection into mice, polyplexes with higher CHE introduction ratios improved protein expression efficiency in the draining lymph nodes and induced robust germinal center responses. Consequently, antibody responses were enhanced with higher CHE introduction ratios in vaccinations targeting a model antigen and the SARS-CoV-2 spike protein. Furthermore, the vaccination elicited both CD8-positive and CD4-positive T cells in a Th1-skewed manner. In a distribution analysis, protein expression from the delivered mRNA was localized to the injection site and draining lymph nodes, avoiding a safety concern associated with systemic distribution. Collectively, this study demonstrates that the introduction of hydrophobic moieties into polyaspartamide derivatives is an effective and safe strategy to enhance the efficacy of polyplex-based mRNA vaccines targeting infectious diseases.
2025
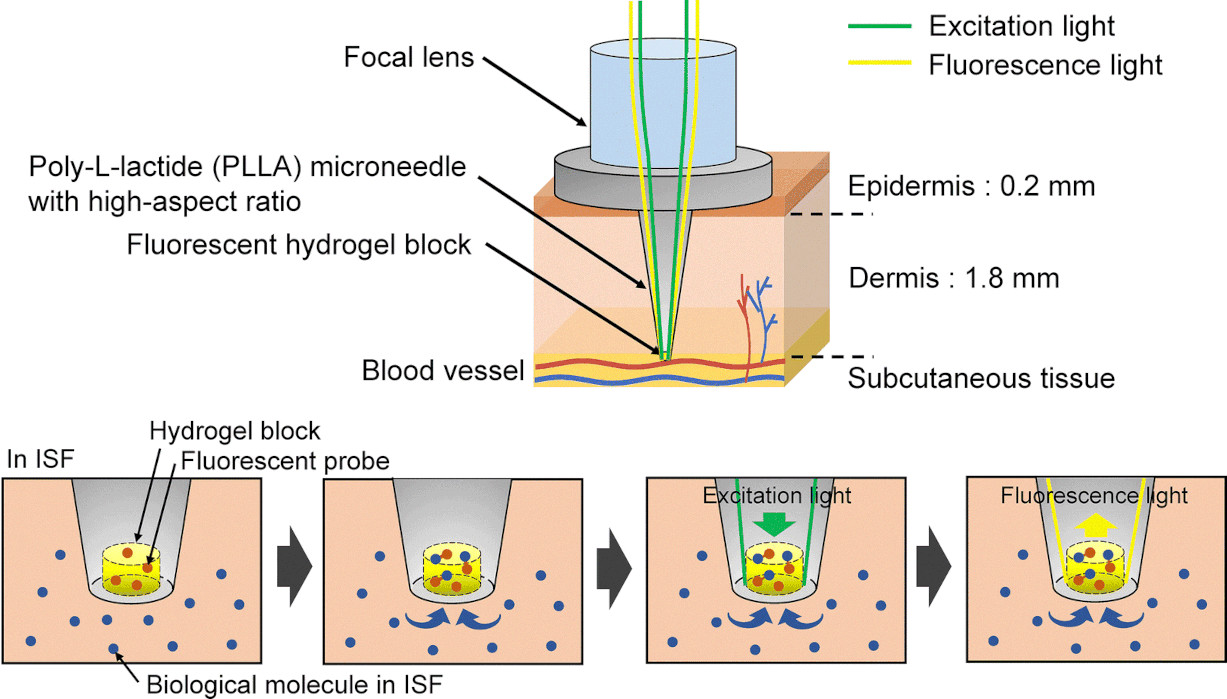 Development of an optical microneedle device embedding sub-nanoliter volumes of boronic acid-based fluorescent hydrogel
Development of an optical microneedle device embedding sub-nanoliter volumes of boronic acid-based fluorescent hydrogel
Fukuhara, M.; Takehara, H.; Barthelmes, K.; Kersch-Hunt, B.; Gardiner, J. E.; Kanda, Y.; Matsumoto, A.; James, T. D.; Ichiki, T.
J. Mater. Chem. B. 2025, Advance Article.
Abstract/Cover Press release Although the diagnostic utility of interstitial fluid (ISF) has been extensively explored, collecting and sensing ISF remains challenging. Microneedle devices offer a promising approach as a minimally invasive method to obtain ISF in a small volume or to sense biomolecules in the ISF within the body. However, conventional enzymatic measurements consume target molecules, compromising sensing reliability, especially in a small volume. To overcome the above issue, we developed fluorescence-based optical microneedles for non-consumptive molecular sensing within tiny ISF samples. The optical microneedle functionalized with a small-volume fluorescent hydrogel block at its tip. The hydrogel block measured 100 µm in diameter and 100 µm in length, with a total volume of 0.79 nL. The microneedle made of amorphous poly-L-lactide (PLLA) had a high-aspect ratio shape (500 µm in base diameter, 200 µm in top diameter, and 2 mm in length), reaching the ISF near the vascular plexus in the reticular layer. In addition, the fluorescent hydrogel was functionalized with boronic acid, which reversibly binds to D-glucose. As proof of our technology, we conducted D-glucose sensing using an optical microneedle. The average value of measurement errors from actual D-glucose concentrations was calculated to be 5.6% in the range of 6.1 to 37.5 mM. Therefore, it was confirmed that the microneedle device is useful for ISF measurements.
Although the diagnostic utility of interstitial fluid (ISF) has been extensively explored, collecting and sensing ISF remains challenging. Microneedle devices offer a promising approach as a minimally invasive method to obtain ISF in a small volume or to sense biomolecules in the ISF within the body. However, conventional enzymatic measurements consume target molecules, compromising sensing reliability, especially in a small volume. To overcome the above issue, we developed fluorescence-based optical microneedles for non-consumptive molecular sensing within tiny ISF samples. The optical microneedle functionalized with a small-volume fluorescent hydrogel block at its tip. The hydrogel block measured 100 µm in diameter and 100 µm in length, with a total volume of 0.79 nL. The microneedle made of amorphous poly-L-lactide (PLLA) had a high-aspect ratio shape (500 µm in base diameter, 200 µm in top diameter, and 2 mm in length), reaching the ISF near the vascular plexus in the reticular layer. In addition, the fluorescent hydrogel was functionalized with boronic acid, which reversibly binds to D-glucose. As proof of our technology, we conducted D-glucose sensing using an optical microneedle. The average value of measurement errors from actual D-glucose concentrations was calculated to be 5.6% in the range of 6.1 to 37.5 mM. Therefore, it was confirmed that the microneedle device is useful for ISF measurements.
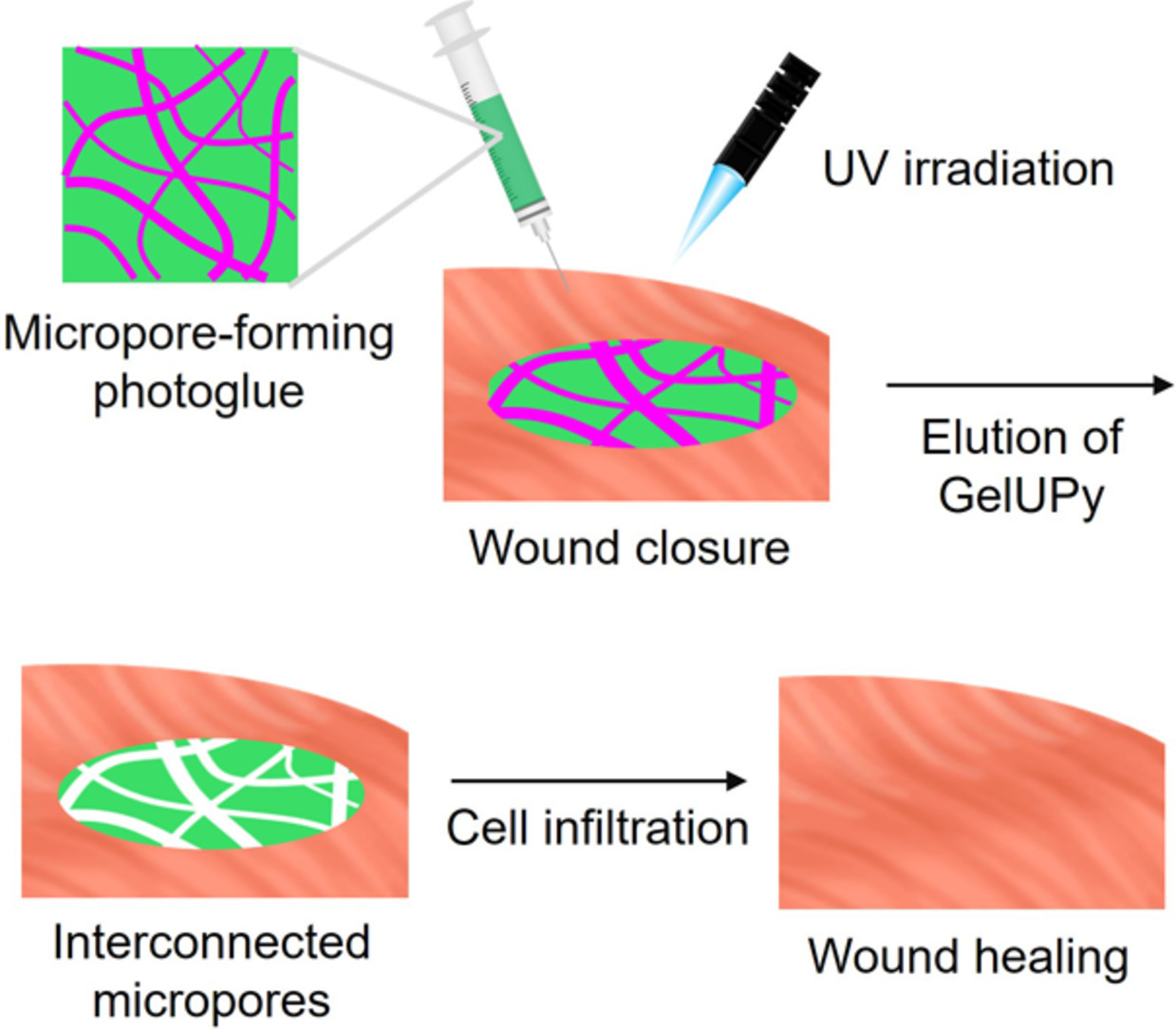 Micropore-forming photocurable tissue adhesive promotes cell infiltration for wound healing
Micropore-forming photocurable tissue adhesive promotes cell infiltration for wound healing
Nishiguchi, A.; Ohta, M.; Palai, D.; Yasue, H.; Sarkar, P.; Komatsu, H.; Taguchi, T.
Acta Biomater. 2025, 206, 215-224.
AbstractTissue adhesives suffer from a trade-off relationship between tissue adhesion strength for long-term wound closure and degradation rate for tissue regeneration, which results in the suppression of postoperative wound healing. Here, we report the development of micropore-forming tissue adhesives with an enhanced cell infiltration capacity for tissue regeneration. By leveraging the phase-separation behavior of gelatin modified with hydrogen-bonding moieties, bicontinuous and micropore-forming photocrosslinked hydrogels were developed. The photocrosslinked hydrogels are injectable and enzymatically degradable, showing high tissue adhesive strength against tissues of the collagen membrane, heart, stomach, and large intestine. Moreover, the microporous structure of the hydrogels could enhance fibroblast infiltration through the micropores. These hydrogels could also induce hair follicle regeneration and wound healing in skin incision wound models. This tissue adhesive has enormous potential for promoting wound healing and preventing postoperative complications.Local administration of an anti-cancer drug using tissue adhesive microparticles based on hydrophobically modified Alaska pollock gelatin for post-surgical cancer chemotherapy
Ito, S.; Komatsu, H.; Minamisakamoto, S.; Palai, D.; Nishiguchi, A.; Taguchi, T.
Colloids Surf. B. Biointerfaces 2025, 254, 114825.
AbstractDespite using endoscopic submucosal dissection to remove tumor tissues or cells, disease recurrence can sometimes occur from residual cancer cells. Although chemotherapy using anti-cancer drugs has been the gold standard, the systemic effects of the drug can lead to side effects, emphasizing the importance of local cancer treatment. However, local administration of anti-cancer drugs to the wet gastrointestinal tissue has been challenging. In this study, we developed a mixed powder composed of paclitaxel microcrystals (PTXMCs) and tissue adhesive microparticles using hydrophobically modified Alaska pollock gelatin microparticles (C10MP), named PTX/C10MP. After hydration for colloidal gel formation, the PTX/C10MP colloidal gel exhibited high tissue adhesion strength and underwater stability to cover gastrointestinal tissues. By immersing this colloidal gel in phosphate buffer solution, PTXMCs were continuously released for up to 18 days. In vivo colon cancer-bearing mouse model experiments revealed that using the PTX/C10MP colloidal gel significantly suppressed cancer growth at 12 days: 1259 ± 135 and 470 ± 151 mm3 for untreated and treated group and improved the survival probability at 18 days: 33 % and 100 % for untreated and treated group. Therefore, our data demonstrate that PTX/C10MP can adhere to the desired gastrointestinal tissue and enable local administration of an anti-cancer drug to kill residual cancer cells following gastrointestinal surgery.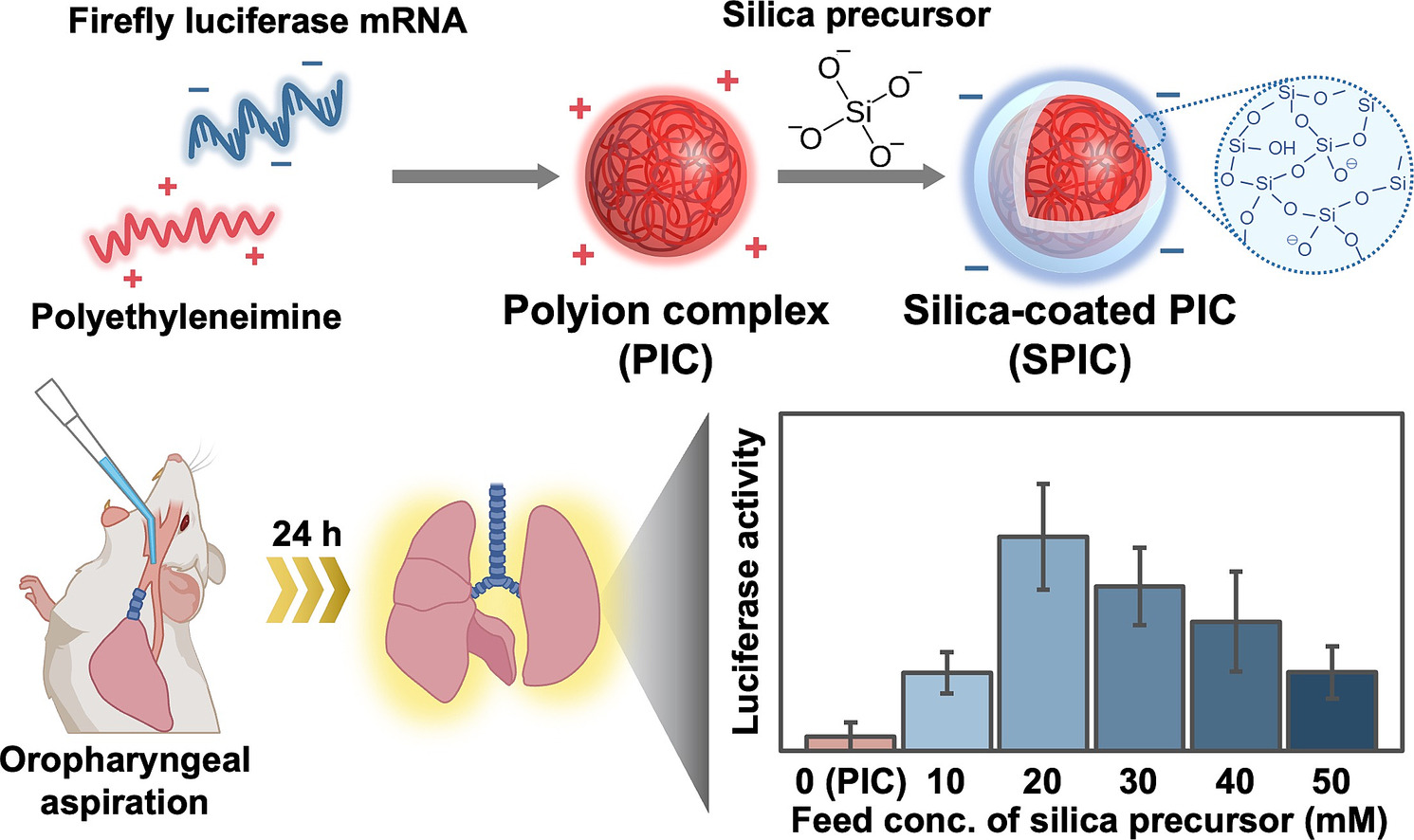 Silica-coated polyion complexes for enhanced pulmonary messenger RNA delivery with optimized silica layer stability
Silica-coated polyion complexes for enhanced pulmonary messenger RNA delivery with optimized silica layer stability
Chang, H.; Aulia, F.; Kamegawa, R.; Qiao, N.; Hori, M.; Ogura, S.; Lau, C. Y. J.; Yamada, N.; Taniwaki, K.; Naito, M.; Kim, H. J.; Miyata, K.
J. Control. Release 2025, 386, 114136.
AbstractPulmonary administration of messenger RNA (mRNA) therapeutics holds great promise for the treatment of lung diseases, as it facilitates local drug accumulation while reducing systemic side effects. However, the fragile nature and inefficient cellular internalization of mRNA in the respiratory epithelium are inherent bottlenecks to mRNA therapy. Although various nanomedicines, mainly lipid and polymeric nanoparticles, have been explored to protect mRNA payloads, their stability in harsh bioenvironments remains severely limited. This study highlighted silica-coated polyion complexes (SPICs) as reversibly stabilized mRNA vehicles for pulmonary mRNA delivery. A silica shell was formed on the surface of mRNA-loaded polyion complexes (PICs) through silicic acid condensation. The silica content of the shells was modulated by altering the concentration of the silica precursor during preparation. Higher silica content more effectively maintained particle uniformity, including size, in simulated lung fluids, whereas uncoated PICs aggregated immediately. However, the mRNA releasability of SPICs was compromised by an increase in silica content. Accordingly, SPICs with optimized silica content induced efficient mRNA expression in cultured lung cancer cells, which was associated with facilitated cellular uptake of mRNA payloads. Ultimately, the optimized SPICs exhibited an order of magnitude higher mRNA expression in the lungs than the uncoated PIC after administration through murine oropharyngeal aspiration. These results demonstrate that SPICs are a promising platform for local mRNA delivery with enhanced mRNA expression by balancing particle stability for payload protection and release.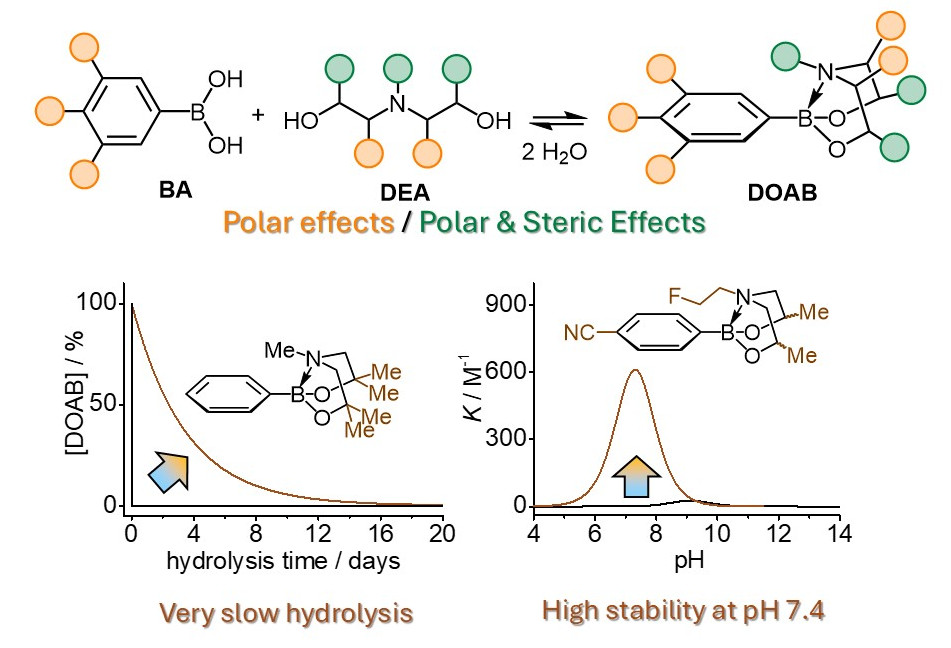 Tuning the Stability and Kinetics of Dioxazaborocanes
Tuning the Stability and Kinetics of Dioxazaborocanes
Barthelmes, K.; Yaginuma, K.; Matsumoto, A.
Chem. Eur. J. 2025, 31(5), e202402625.
Abstract/Cover We investigated the equilibrium reaction of boronic acid (BA), diethanolamines (DEA), and 1,3,6,2-dioxazaborocanes (DOAB) in aqueous solutions, both theoretically and experimentally. Our findings show that the association constant can be adjusted by substituting BA and DEA derivatives, ranging from 100 to 103 M−1, exhibiting a bell-shaped pH dependency. The highest stability was achieved when the pKa values of DEA and BA were closely matched. This approach enabled the preparation of a highly stable DOAB under physiological conditions. Furthermore, the hydrolysis kinetics of DOABs were controllable over a range of five orders of magnitude based on the substituent’s steric effect. In the slowest case, this resulted in quasi-static stability with only 1 % cleavage in the first hour, followed by a week-long cleavage period to reach equilibrium. These insights could establish a unique chemistry platform for designing scheduled cleavability on a day-to-week timescale, relevant to protein engineering, immunotherapy, and other smart drug delivery applications.
We investigated the equilibrium reaction of boronic acid (BA), diethanolamines (DEA), and 1,3,6,2-dioxazaborocanes (DOAB) in aqueous solutions, both theoretically and experimentally. Our findings show that the association constant can be adjusted by substituting BA and DEA derivatives, ranging from 100 to 103 M−1, exhibiting a bell-shaped pH dependency. The highest stability was achieved when the pKa values of DEA and BA were closely matched. This approach enabled the preparation of a highly stable DOAB under physiological conditions. Furthermore, the hydrolysis kinetics of DOABs were controllable over a range of five orders of magnitude based on the substituent’s steric effect. In the slowest case, this resulted in quasi-static stability with only 1 % cleavage in the first hour, followed by a week-long cleavage period to reach equilibrium. These insights could establish a unique chemistry platform for designing scheduled cleavability on a day-to-week timescale, relevant to protein engineering, immunotherapy, and other smart drug delivery applications.
2024
Increased mechanical stability and permeability by filling the interconnected pores of porous microneedles
Barthelmes, K.; Sathitaphiwan, K.; Janwimaluang, N.; Ikehara, K.; Matsumoto, A.
Jpn. J. Appl. Phys. 2024, 63(2).
AbstractA new polymeric microneedle (MN) fabrication technique is described in order to facilitate both higher mechanical stability and continuous drug release capability, a well-recognized challenge in the community. The technique involves filling the pores of a porous MN (PMN) array with a hydrogel. Cellulose acetate (CA) was used to prepare PMN, the interconnected cavity of which was then occupied by a crosslinked poly(N-isopropylacrylamide) hydrogel. Alkali treatment of the PMN array resulted in deacetylation of CA and improved the hydrophilicity on the surface. The hydrogel was readily incorporated by thermal polymerization of the monomers soaked to the PMN array. Mechanical strength tests demonstrated that pore filling enhanced the PMN stability by up to 50%, which was well-above the threshold required for skin penetration. The permeability of the hydrogel remained after pore filling and the drug release rate could be varied by alkali treatment process.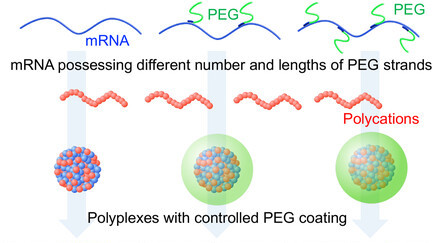 Poly(ethylene Glycol) (PEG)–OligoRNA Hybridization to mRNA Enables Fine-Tuned Polyplex PEGylation for Spleen-Targeted mRNA Delivery
Poly(ethylene Glycol) (PEG)–OligoRNA Hybridization to mRNA Enables Fine-Tuned Polyplex PEGylation for Spleen-Targeted mRNA Delivery
Suzuki, M.; Mochida, Y.; Hori, M.; Hayashi, A.; Toh, K.; Tockary, T. A.; Liu, X.; Marx, V.; Yokoo, H.; Miyata, K.; Oba, M.; Uchida, S.
Small Science 2024, 4(4), 2300258.
AbstractOrgan-selective targeting of mRNA polyplexes has been rarely explored despite the substantial potential of polymer-based systems in mRNA delivery. In this study, spleen-selective delivery of polyplexes is achieved by employing mRNA engineering to coat them with poly(ethylene glycol) (PEG). In this approach, mRNA is hybridized with PEGylated complementary RNA oligonucleotides (PEG–OligoRNAs), followed by the addition of linear poly(ethyleneimine). In this method, it is ensured that nearly all added PEG strands bind to the polyplexes, thereby enabling precise control of PEG amounts on the surface. Following systemic injection into mice, non-PEGylated polyplexes yield robust protein expression in the lung and spleen. Intriguingly, adding a small number of PEG–OligoRNAs drastically reduces protein expression efficiency in the lung while preserving it in the spleen, realizing spleen targeting of mRNA polyplexes. Furthermore, PEGylated polyplexes demonstrate their potential utility in mRNA vaccination. In mechanistic analyses, non-PEGylated polyplexes immediately agglomerate in the blood and deposit in the lung. Coating polyplexes with a small amount of short PEG effectively prevents these processes. Notably, even slight changes in PEG amounts and lengths dramatically impact the physicochemical properties and biological functionalities of the polyplexes, emphasizing the benefits of an mRNA engineering-based approach for fine-tuning polyplex PEG coating.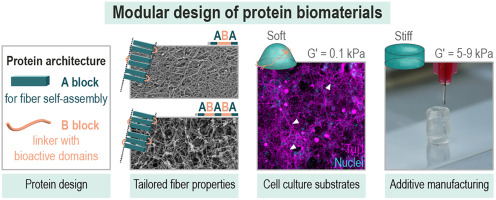 Designed modular protein hydrogels for biofabrication
Designed modular protein hydrogels for biofabrication
Dranseike, D.; Ota, Y.; Edwardson, T. G. W.; Guzzi, E. A.; Hori, M.; Nakic, Z. R.; Deshmukh, D. V.; Levasseur, M. D.; Mattli, K.; Tringides, C. M.; Zhou, J.; Hilvert, D.; Peters, C.; Tibbitt, M. W.
Acta Biomater. 2024, 177, 107-117.
AbstractDesigning proteins that fold and assemble over different length scales provides a way to tailor the mechanical properties and biological performance of hydrogels. In this study, we designed modular proteins that self-assemble into fibrillar networks and, as a result, form hydrogel materials with novel properties. We incorporated distinct functionalities by connecting separate self-assembling (A block) and cell-binding (B block) domains into single macromolecules. The number of self-assembling domains affects the rigidity of the fibers and the final storage modulus G′ of the materials. The mechanical properties of the hydrogels could be tuned over a broad range (G′ = 0.1 – 10 kPa), making them suitable for the cultivation and differentiation of multiple cell types, including cortical neurons and human mesenchymal stem cells. Moreover, we confirmed the bioavailability of cell attachment domains in the hydrogels that can be further tailored for specific cell types or other biological applications. Finally, we demonstrate the versatility of the designed proteins for application in biofabrication as 3D scaffolds that support cell growth and guide their function.Functionalization of Poly-L-Lactic Acid Microneedle Tips using Hydrogel Photopolymerization
Fukuhara, M.; Takehara, H.; Kanda, Y.; Matsumoto, A.; Ichiki, T.
J. Photopolym. Sci. Tec. 2024, 37(2), 233-237.
AbstractAlthough the diagnostic utility of interstitial fluid (ISF) has been extensively explored, collecting and sensing ISF in the body remains challenging. Microneedle devices offer a promising approach for minimally invasive methods to obtain ISF with a small volume or to sense biomolecules in the ISF within the body. In this study, we developed a microneedle with a small volume hydrogel block at the needle tip. The hydrogel block was formed through a photopolymerization reaction of polyethylene glycol diacrylate (PEGDA) under 365 nm irradiation. The hydrogel block measured 100 µm in diameter and 100 µm in length, with a total volume of 0.79 nL. The fabricated microneedle had a high-aspect ratio shape (500 µm in base diameter, 200 µm in top diameter, and 2000 µm in length), reaching the reticular layer containing blood vessels. Therefore, microneedle devices can be utilized to obtain ISF with a small volume and to sense biomolecules within the body.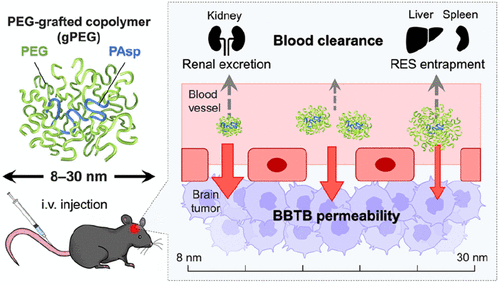 Size-Dependent Glioblastoma Targeting by Polymeric Nanoruler with Prolonged Blood Circulation
Size-Dependent Glioblastoma Targeting by Polymeric Nanoruler with Prolonged Blood Circulation
Ishibashi, Y.; Naito, M.; Watanuki, Y.; Hori, M.; Ogura, S.; Taniwaki, K.; Cho, M.; Komiya, R.; Mochida, Y.; Miyata, K.
Bioconjugate Chem. 2024, 35(8), 1154-1159.
AbstractCurrently, there is no effective treatment for glioblastoma multiforme (GBM), the most frequent and malignant type of brain tumor. The blood–brain (tumor) barrier (BB(T)B), which is composed of tightly connected endothelial cells and pericytes (with partial vasculature collapse), hampers nanomedicine accumulation in tumor tissues. We aimed to explore the effect of nanomedicine size on passive targeting of GBM. A series of size-tunable poly(ethylene glycol) (PEG)-grafted copolymers (gPEGs) were constructed with hydrodynamic diameters of 8–30 nm. Biodistribution studies using orthotopic brain tumor-bearing mice revealed that gPEG brain tumor accumulation was maximized at 10 nm with ∼14 dose %/g of tumor, which was 19 times higher than that in the normal brain region and 4.2 times higher than that of 30-nm gPEG. Notably, 10-nm gPEG exhibited substantially higher brain tumor accumulation than 11-nm linear PEG owing to the prolonged blood circulation property of gPEGs, which is derived from a densely PEG-packed structure. 10 nm gPEG exhibited deeper penetration into the brain tumor tissue than the larger gPEGs did (>10 nm). This study demonstrates, for the first time, the great potential of a nanomedicine downsizing strategy for passive GBM targeting.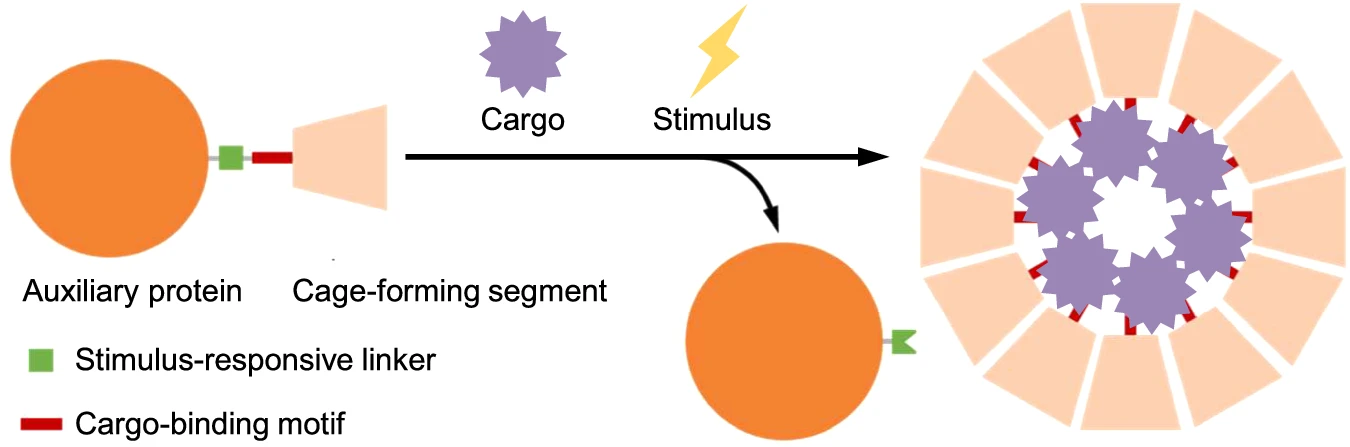 Stimulus-responsive assembly of nonviral nucleocapsids
Stimulus-responsive assembly of nonviral nucleocapsids
Hori, M.; Steinauer, A.; Tetter, S.; Hälg, J.; Manz, E.-M.; Hilvert, D.
Nat. Commun. 2024, 15(1), 3576.
AbstractControlled assembly of a protein shell around a viral genome is a key step in the life cycle of many viruses. Here we report a strategy for regulating the co-assembly of nonviral proteins and nucleic acids into highly ordered nucleocapsids in vitro. By fusing maltose binding protein to the subunits of NC-4, an engineered protein cage that encapsulates its own encoding mRNA, we successfully blocked spontaneous capsid assembly, allowing isolation of the individual monomers in soluble form. To initiate RNA-templated nucleocapsid formation, the steric block can be simply removed by selective proteolysis. Analyses by transmission and cryo-electron microscopy confirmed that the resulting assemblies are structurally identical to their RNA-containing counterparts produced in vivo. Enzymatically triggered cage formation broadens the range of RNA molecules that can be encapsulated by NC-4, provides unique opportunities to study the co-assembly of capsid and cargo, and could be useful for studying other nonviral and viral assemblies.
Reviews
ボロン酸高分子ネットワークバイオ材料の設計と医療応用 (Design and medical application of boronic acid polymer network biomaterials)
松元 亮.
ネットワークポリマー論文集 (Network Polymer Papers) 2024, 45(1), 44-54.
Abstractボロン酸は,水中で様々な生体分子と可逆的な結合性を呈する。糖との結合性から「ボロノレクチン」とも呼ばれる。その結合強度や選択性は合成化学的に可変である。ボロン酸による可逆的な分子認識においては,それ自身の解離状態と同期した顕著な親疎水性変化が付随し,これらを適切に分子デザインすることで,「ネットワークポリマー」の複合的・階層的な環境応答性の付与が可能となる。本稿では,筆者らが展開する「ボロノレクチン」の機能を応用した疾病診断やドラッグデリバリーシステムに関する研究から,特に,細胞内環境応答的な核酸医薬の送達システム,シアル酸認識によるがん診断及び標的治療技術,糖尿病治療を目的としたグルコース応答型インスリン供給システム等について概説する。
Books
Wearable Artificial Pancreas Device Technology
Matsumoto, A.
Wearable Biosensing in Medicine and Healthcare 2024, 249-266 (Springer).
AbstractThis chapter covers the development of closed-loop insulin delivery systems known as artificial pancreas systems (APSs). These systems can be either electronics-based or electronics-free, and there is a continuous drive to make them both wearable and user-friendly. First, we outline the development and validation status of electronics-based APSs. Then, we summarize the growing research effort to develop electronics-free, chemically-controlled APSs, with particular emphasis on ongoing efforts to make them wearable. We also discuss current challenges, possible solutions, and future perspectives based on promising results from a recent clinical trial.貼るだけ人工膵臓デバイスの開発 (Development of a stick-on ’artificial pancreas’ device)
松元 亮; 松本 裕子; 金井 紗綾香; Barthelmes, K.; 堀 真緒; 田中 都; 伊藤 美智子; 菅波 孝祥.
血糖測定・コントロールの最新動向 (Latest Trends in Blood Glucose Monitoring and Control) 2024, Chapter 5.4, (CMC Publishing Co., Ltd).
Drug Delivery Systems for Muscles
Naito, M.; Hori, M.
Drug Delivery Systems for Musculoskeletal Tissues 2024, 189-208 (Springer).
AbstractMuscle diseases and disorders can cause a loss of function and mobility, significantly reducing the quality of life for patients and their caregivers. The treatment options for muscle diseases and disorders are limited or, in most cases, unavailable. One of the barriers to the development of novel treatment modalities is the low diffusion rate of drugs from blood to muscle, known as the “blood-muscle barrier.” Therefore, drug delivery systems and strategies targeting muscles are essential for the effective treatment of muscle diseases and disorders. This chapter provides a comprehensive overview of muscle-targeted drugs and their delivery strategies to the musculoskeletal system. Fundamental information on the structure and molecular biology of muscles is also described to help further our understanding of the existing muscle-targeted drug delivery systems.
2023
 Block catiomers with flanking hydrolyzable tyrosinate groups enhance in vivo mRNA delivery via π-π stacking-assisted micellar
assembly
Block catiomers with flanking hydrolyzable tyrosinate groups enhance in vivo mRNA delivery via π-π stacking-assisted micellar
assembly
Yang, W.; Miyazaki, T.; Nakagawa, Y.; Boonstra, E.; Masuda, K.; Nakashima, Y.; Chen, P.; Mixich, L.; Barthelmes, K.; Matsumoto, A.; Mi, P.; Uchida, S.; Cabral, H.
Sci. Technol. Adv. Mater. 2023, 24(1).
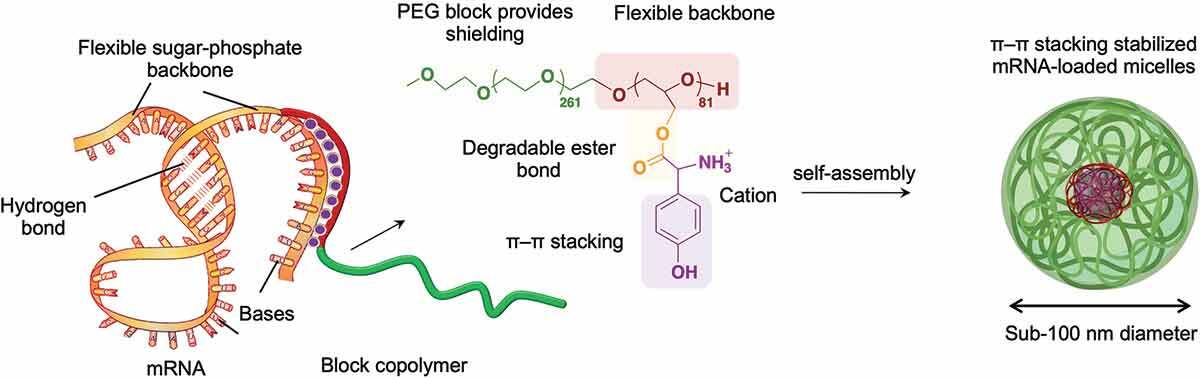
AbstractMessenger RNA (mRNA) therapeutics have recently demonstrated high clinical potential with the accelerated approval of SARS-CoV-2 vaccines. To fulfill the promise of unprecedented mRNA-based treatments, the development of safe and efficient carriers is still necessary to achieve effective delivery of mRNA. Herein, we prepared mRNA-loaded nanocarriers for enhanced in vivo delivery using biocompatible block copolymers having functional amino acid moieties for tunable interaction with mRNA. The block copolymers were based on flexible poly(ethylene glycol)-poly(glycerol) (PEG-PG) modified with glycine (Gly), leucine (Leu) or tyrosine (Tyr) via ester bonds to generate block catiomers. Moreover, the amino acids can be gradually detached from the block copolymers after ester bond hydrolyzation, avoiding cytotoxic effects. When mixed with mRNA, the block catiomers formed narrowly distributed polymeric micelles with high stability and enhanced delivery efficiency. Particularly, the micelles based on tyrosine-modified PEG-PG (PEG-PGTyr), which formed a polyion complex (PIC) and pi-pi stacking with mRNA, displayed excellent stability against polyanions and promoted mRNA integrity in serum. PEG-PGTyr-based micelles also increased the cellular uptake and the endosomal escape, promoting high protein expression both in vitro and in vivo. Furthermore, the PEG-PGTyr-based micelles significantly extended the half-life of the loaded mRNA after intravenous injection. Our results highlight the potential of PEG-PGTyr-based micelles as safe and effective carriers for mRNA, expediting the rational design of polymeric materials for enhanced mRNA delivery.Lysosomal cholesterol overload in macrophages promotes liver fibrosis in a mouse model of NASH
Itoh, M.; Tamura, A.; Kanai, S.; Tanaka, M.; Kanamori, Y.; Shirakawa, I.; Ito, A.; Oka, Y.; Hidaka, I.; Takami, T.; Honda, Y.; Maeda, M.; Saito, Y.; Murata, Y.; Matozaki, T.; Nakajima, A.; Kataoka, Y.; Ogi, T.; Ogawa, Y.; Suganami, T.
J. Exp. Med. 2023, 220(11).
AbstractLysosomal cholesterol overload triggers phenotypic changes and profibrotic activation of macrophages around dead hepatocytes containing cholesterol crystals in the development of NASH. Cholesterol excretion from macrophages using & beta;-cyclodextrin polyrotaxane, which is designed to release & beta;-cyclodextrin in lysosomes, alleviates liver fibrosis. Accumulation of lipotoxic lipids, such as free cholesterol, induces hepatocyte death and subsequent inflammation and fibrosis in the pathogenesis of nonalcoholic steatohepatitis (NASH). However, the underlying mechanisms remain unclear. We have previously reported that hepatocyte death locally induces phenotypic changes in the macrophages surrounding the corpse and remnant lipids, thereby promoting liver fibrosis in a murine model of NASH. Here, we demonstrated that lysosomal cholesterol overload triggers lysosomal dysfunction and profibrotic activation of macrophages during the development of NASH. & beta;-cyclodextrin polyrotaxane (& beta;CD-PRX), a unique supramolecule, is designed to elicit free cholesterol from lysosomes. Treatment with & beta;CD-PRX ameliorated cholesterol accumulation and profibrotic activation of macrophages surrounding dead hepatocytes with cholesterol crystals, thereby suppressing liver fibrosis in a NASH model, without affecting the hepatic cholesterol levels. In vitro experiments revealed that cholesterol-induced lysosomal stress triggered profibrotic activation in macrophages predisposed to the steatotic microenvironment. This study provides evidence that dysregulated cholesterol metabolism in macrophages would be a novel mechanism of NASH.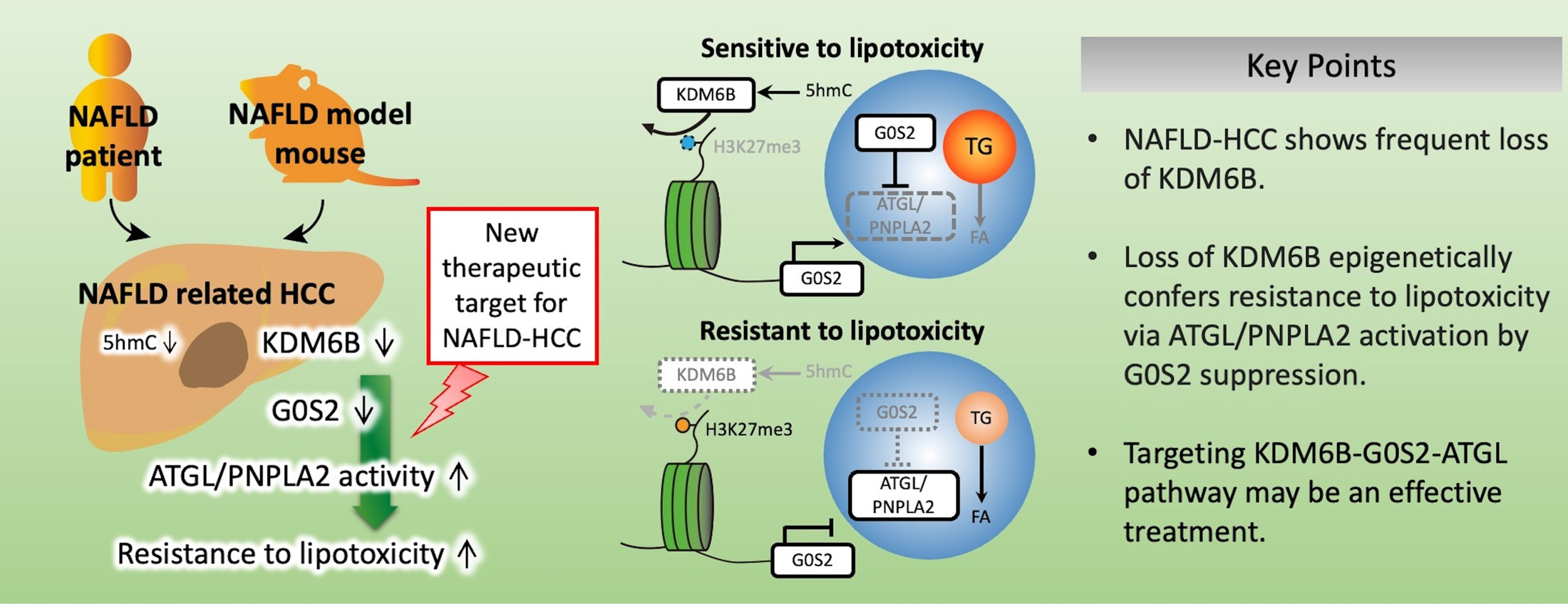 Loss of KDM6B epigenetically confers resistance to lipotoxicity in nonalcoholic fatty liver disease-related HCC
Loss of KDM6B epigenetically confers resistance to lipotoxicity in nonalcoholic fatty liver disease-related HCC
Hatano, M.; Akiyama, Y.; Shimada, S.; Yagi, K.; Akahoshi, K.; Itoh, M.; Tanabe, M.; Ogawa, Y.; Tanaka, S.
Hepatol. Commun. 2023, 7(10), e0277.
AbstractBackground: NAFLD caused by abnormalities in hepatic lipid metabolism is associated with an increased risk of developing HCC. The molecular mechanisms underlying the progression of NAFLD-related HCC are not fully understood. We investigated the molecular mechanism and role of KDM6B downregulation in NAFLD-related HCC after the KDM6B gene was identified using microarray analysis as commonly downregulated in mouse NAFLD-related HCC and human nonhepatitis B and nonhepatitis C viral-HCC.Methods: The 5-hydroxymethylcytosine levels of KDM6B in HCC cells were determined using glycosylated hydroxymethyl-sensitive PCR. Microarray and chromatin immunoprecipitation analyses using KDM6B-knockout (KO) cells were used to identify KDM6B target genes. Lipotoxicity was assessed using a palmitate-treated cell proliferation assay. Immunohistochemistry was used to evaluate KDM6B expression in human HCC tissues.Results: KDM6B expression levels in HCC cells correlated with the 5-hydroxymethylcytosine levels in the KDM6B gene body region. Gene set enrichment analysis revealed that the lipid metabolism pathway was suppressed in KDM6B-KO cells. KDM6B-KO cells acquired resistance to lipotoxicity (p < 0.01) and downregulated the expression of G0S2, an adipose triglyceride lipase/patatin like phospholipase domain containing 2 (ATGL/PNPLA2) inhibitor, through increased histone H3 lysine-27 trimethylation levels. G0S2 knockdown in KDM6B-expressed HCC cells conferred lipotoxicity resistance, whereas ATGL/PNPLA2 inhibition in the KDM6B-KO cells reduced these effects. Immunohistochemistry revealed that KDM6B expression was decreased in human NAFLD-related HCC tissues (p < 0.001), which was significantly associated with decreased G0S2 expression (p = 0.032).Conclusions: KDM6B-disrupted HCC acquires resistance to lipotoxicity via ATGL/PNPLA2 activation caused by epigenetic downregulation of G0S2 expression. Reduced KDM6B and G0S2 expression levels are common in NAFLD-related HCC. Targeting the KDM6B-G0S2-ATGL/PNPLA2 pathway may be a useful therapeutic strategy for NAFLD-related HCC.
Reviews
"Borono-lectin"-mediated Crosstalk and Its Application to Bioengineering
Matsumoto, A.
Yakugaku Zasshi-J. Pharm. Soc. Jpn. 2023, 143(5), 435-441.
AbstractBoronic acids are able to reversibly interact with the diol groups, a commonly found motif in biomolecules including sugars, ribose and catechols. For their carbohydrate-binding properties, they can be regarded as a synthetic mimic of lectins and often termed as “borono-lectins.” Importantly, the borono-lectins platform can be chemically tailored to manifest a broad profile of binding strength and specificity. Besides the structural versatility, some derivatives can undergo a sharp inversion in the state of hydration in synchronization with the molecular recognitions. This feature, when combined with amphiphilic polymeric backbones, translates into many creative principles for fine-tuning or switching the hydration and more complex molecular assemblies in a way interactive with biology. Here we provide a brief overview of our recent efforts on the related applications with special focuses on smart insulin delivery systems and sialic acid detections relevant to cancer diagnosis and treatment among others.’ボロノレクチン’を応用した糖鎖シアル酸検出と腫瘍ターゲティング (Sugar chain sialic acid detection and tumor targeting using ’boronorectin’)
松元 亮.
バイオマテリアル (Journal of Japanese Society for Biomaterials) 2023, 42(1), 92-93.
Books
もっとよくわかる!線維化と疾患 (Understand more! Fibrosis and disease)
菅波 孝祥; 田中 都; 伊藤 美智子.
実験医学別冊 もっとよくわかる!シリーズ (Experimental Medicine Special Edition: Understand more! series) 2023, (羊土社).
Abstract炎症や疾患研究をしていると必ず通る「線維化」ですが,その進展機構や実態を正しく理解できていますか?メカニズムと病態の両面からわかりやすく解説し,最新論文を読む前の学びはじめに最適な初の入門書が登場!
2022
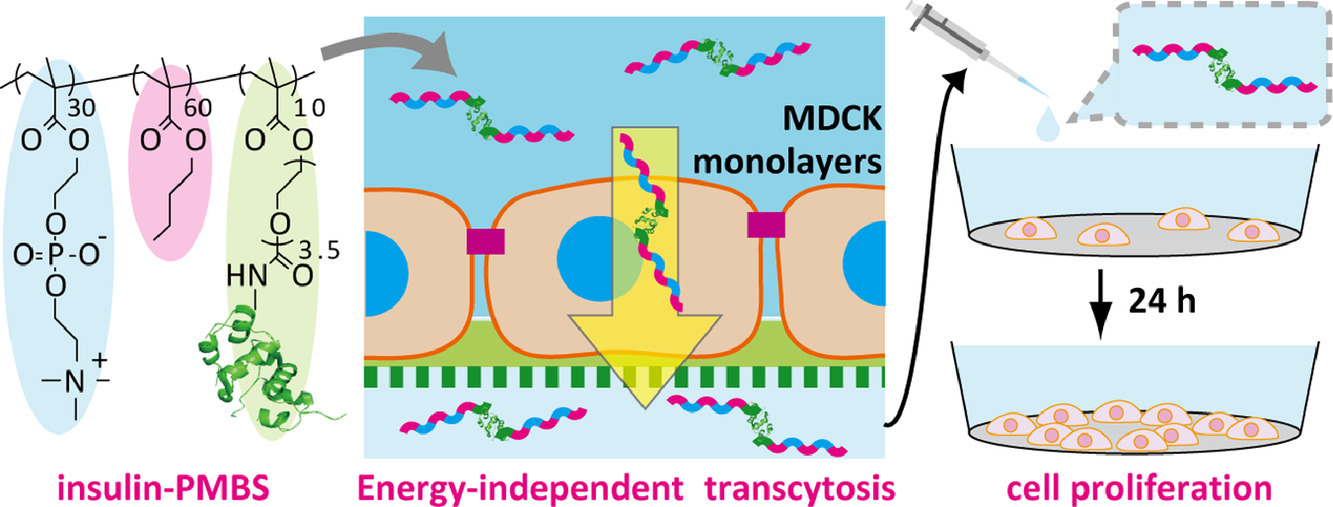 Transepithelial delivery of insulin conjugated with phospholipid-mimicking polymers via biomembrane fusion-mediated transcellular pathways
Transepithelial delivery of insulin conjugated with phospholipid-mimicking polymers via biomembrane fusion-mediated transcellular pathways
Hatano, H.; Meng, F.; Sakata, M.; Matsumoto, A.; Ishihara, K.; Miyahara, Y.; Goda, T.
Acta Biomater. 2022, 140, 674-685.
AbstractEpithelial barriers that seal cell gaps by forming tight junctions to prevent the free permeation of nutrients, electrolytes, and drugs, are essential for maintaining homeostasis in multicellular organisms. The development of nanocarriers that can permeate epithelial tissues without compromising barrier function is key for establishing a safe and efficient drug delivery system (DDS). Previously, we have demonstrated that a water-soluble phospholipid-mimicking random copolymer, poly(2-methacryloyloxyethyl phosphorylcholine30- random n -butyl methacrylate70) (PMB30W), enters the cytoplasm of live cells by passive diffusion manners, without damaging the cell membranes. The internalization mechanism was confirmed to be amphiphilicity-induced membrane fusion. In the present study, we demonstrated energy independent permeation of PMB30W through the model epithelial barriers of Madin-Darby canine kidney (MDCK) cell monolayers in vitro . The polymer penetrated epithelial MDCK monolayers via transcellular pathways without breaching the barrier functions. This was confirmed by our unique assay that can monitor the leakage of the proton as the smallest indicator across the epithelial barriers. Moreover, energy independent transepithelial permeation was achieved when insulin was chemically conjugated with the phospholipid-mimicking nanocarrier. The bioactivity of insulin as a growth factor was found to be maintained even after translocation. These fundamental findings may aid the establishment of transepithelial DDS with advanced drug efficiency and safety.Statement of significanceA nanocarrier that can freely permeate epithelial tissues without compromising barrier function is key for successful DDS. Existing strategies mainly rely on paracellular transport associated with tight junction breakdown or transcellular transport via transporter recognition-mediated active uptake. These approaches raise concerns about efficiency and safety. In this study, we performed non-endocytic permeation of phospholipid-mimicking polymers through the model epithelial barriers in vitro . The polymer penetrated via transcytotic pathways without breaching the barriers of biomembrane and tight junction. Moreover, transepithelial permeation occurred when insulin was covalently attached to the nanocarrier. The bioactivity of insulin was maintained even after translocation. The biomimetic design of nanocarrier may realize safe and efficient transepithelial DDS.& nbsp;(c) 2021 Acta Materialia Inc. Published by Elsevier Ltd. All rights reserved.A Porous Reservoir-Backed Boronate Gel Microneedle for Efficient Skin Penetration and Sustained Glucose-Responsive Insulin Delivery
Chen, S.; Miyazaki, T.; Itoh, M.; Matsumoto, H.; Moro-oka, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.; Matsumoto, A.
Gels 2022, 8(2), 74.
AbstractRecently, phenylboronic acid (PBA) gel containing microneedle (MN) technology with acute and sustained glucose-sensitive functionality has attracted significant research attention. Herein, we report a polyvinyl alcohol(PVA)-coated MNs patch with an interconnected porous gel drug reservoir for enhanced skin penetration efficiency and mechanical strength. The hybrid MNs patch fabricated with a novel, efficient method displayed a “cake-like” two-layer structure, with the tip part being composed of boronate-containing smart gel attached to a porous gel layer as a drug reservoir. The porous structure provides the necessary structural support for skin insertion and space for insulin loading. The mechanical strength of the hybrid MNs patch was further enhanced by surface coating with crystallized PVA. Compared with MNs patches attached to hollow drug reservoirs, this hybrid MNs patch with a porous gel reservoir was shown to be able to penetrate the skin more effectively, and is promising for on-demand, long-acting transdermal insulin delivery with increased patient compliance.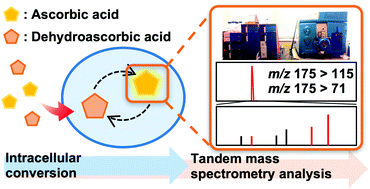 Determination of intracellular ascorbic acid using tandem mass
spectrometry
Determination of intracellular ascorbic acid using tandem mass
spectrometry
Abe, C.; Higuchi, O.; Matsumoto, A.; Miyazawa, T.
Analyst 2022, 147(12), 2640-2643.
AbstractAscorbic acid is involved in a variety of biological events. Nevertheless, its detailed intracellular behavior remains unexplored due to a lack of sensitive analytical methods. Here we report a method using HPLC-ESI-MS/MS achieving the lowest detection limit ever reported (1 pg), to provide mechanistic insight into the intracellular ascorbic acid physiology.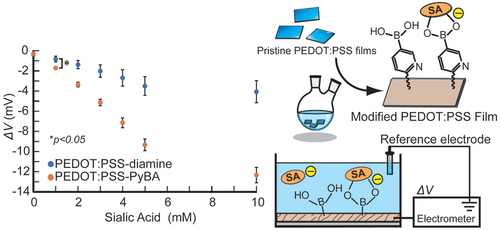 Sialic acid biosensing by post-printing modification of PEDOT:PSS with
pyridylboronic acid
Sialic acid biosensing by post-printing modification of PEDOT:PSS with
pyridylboronic acid
Fujisaki, H.; Matsumoto, A.; Miyahara, Y.; Goda, T.
Sci. Technol. Adv. Mater. 2022, 23(1), 525-534.
AbstractA poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) (PEDOT:PSS)-based conducting polymer, which has biorecognition capabilities, has promising biosensing applications. Previously, we developed a facile method for post-printing chemical modification of PEDOT:PSS thin films from commercial sources. Molecular recognition elements were directly introduced into the PSS side chain by a two-step chemical reaction: introduction of an ethylenediamine linker via an acid chloride reaction of the sulfonate moiety, and subsequent receptor attachment to the linker via amine coupling. In this study, the same method was used to introduce 6-carboxypyridine-3-boronic acid (carboxy-PyBA) into the linker for specifically detecting N-acetylneuraminic acid (sialic acid, SA), as a cancer biomarker. The surface-modified PEDOT:PSS films were characterized by X-ray photoelectron spectroscopy, attenuated total reflection Fourier-transform infrared spectroscopy, and static water contact angle and conductivity measurements. The specific interaction between PyBA and SA was detected by label-free reagent-free potentiometry. The SA-specific negative potential responses of modified PEDOT:PSS electrodes, which was ascribed to an SA carboxyl anion, were observed in a physiologically relevant SA range (1.6-2.9 mM) at pH 5, in a concentration-dependent manner even in the presence of 10% fetal bovine serum. The sensitivity was -2.9 mV/mM in 1-5 mM SA with a limit of detection of 0.7 mM. The sensing performances were almost equivalent to those of existing graphene-based electrical SA sensors. These results show that our chemical derivatization method for printing PEDOT:PSS thin films will have applications in SA clinical diagnostics.
Reviews
 Food Antioxidants and Aging: Theory, Current Evidence and Perspectives
Food Antioxidants and Aging: Theory, Current Evidence and Perspectives
Miyazawa, T.; Abe, C.; Burdeos, G. C.; Matsumoto, A.; Toda, M.
Nutraceuticals 2022, 2(3), 181-204.
AbstractThe concept of food and aging is of great concern to humans. So far, more than 300 theories of aging have been suggested, and approaches based on these principles have been investigated. It has been reported that antioxidants in foods might play a role in human aging. To clarify the current recognition and positioning of the relationship between these food antioxidants and aging, this review is presented in the following order: (1) aging theories, (2) food and aging, and (3) individual food antioxidants and aging. Clarifying the significance of food antioxidants in the field of aging will lead to the development of strategies to achieve healthy human aging.
2021
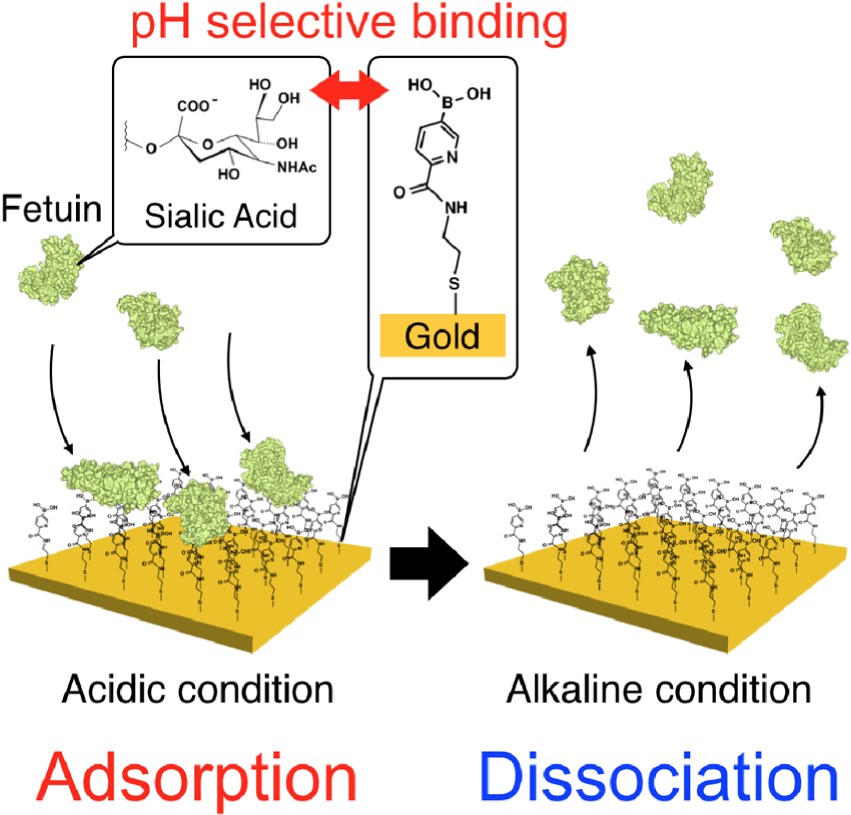 pH-responsive Adsorption and Dissociation of Sialic Acid Expressed
Protein on Boronic Acid Immobilized Surface
pH-responsive Adsorption and Dissociation of Sialic Acid Expressed
Protein on Boronic Acid Immobilized Surface
Horiguchi, Y.; Barthelmes, K.; Miyahara, Y.; Matsumoto, A.
Chem. Lett. 2021, 50(8), 1467-1469.
AbstractDetection and isolation of sialic acid (SA) over-expressed biomolecules is of continuous interest in cancer-related research. Boronic acid-based SA-recognition chemistry provides a synthetic molecular basis for such techniques. 5-Boronopicolinic acid, a derivative with the reportedly highest SA-binding strength, was immobilized to a gold surface and studied for its interaction with fetuin, a blood-circulating glycoprotein implicated in cancers. A submicron-molar level sensitivity was achievable, while the sample could be readily dissociated by modulating the pH.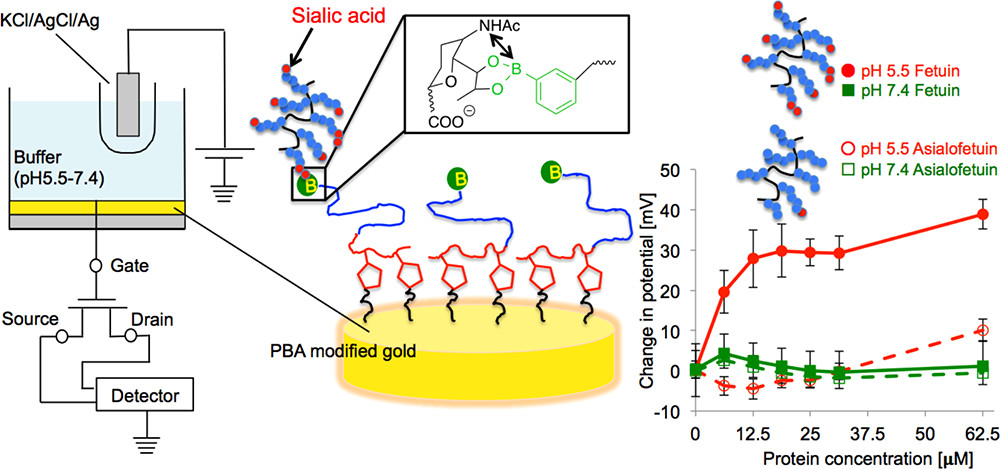 Potentiometric Determination of Circulating Glycoproteins by Boronic
Acid End-Functionalized Poly(ethylene glycol)-Modified Electrode
Potentiometric Determination of Circulating Glycoproteins by Boronic
Acid End-Functionalized Poly(ethylene glycol)-Modified Electrode
Matsumoto, A.; Osawa, S.; Arai, T.; Maejima, Y.; Otsuka, H.; Miyahara, Y.
Bioconjugate Chem. 2021, 32(2), 239-244.
Abstract/Cover Despite tremendous complexity in glycan structure, sialic acid (SA)
provides an analytically accessible index for glycosylation, owing to
its uniquely anionic nature and glycan-chain terminal occupation. Taking
advantage of boronic acid (BA) based SA-recognition chemistry, we here
demonstrate a label-free, no enzymatic, potentiometric determination of
fetuin, a blood-circulating glycoprotein implicated in physiological and
various pathological states. A phenylboronic acid (PBA)
omega-end-functionalized poly(ethylene glycol) (PEG) with an
alpha-tethering unit bearing pendent alkyne groups was “grafted-to”
a gold electrode modified with 11-azide-undecathiol by a
copper-catalyzed azide-alkyne cycloaddition reaction. Using the
electrode, fetuin was potentiometrically detectable with a mu
M-order-sensitivity that is comparable to what is found in
blood-collected specimen. Our finding may have implications for
developing a remarkably economic hemodiagnostic technology with ease of
downsizing and mass production.
Despite tremendous complexity in glycan structure, sialic acid (SA)
provides an analytically accessible index for glycosylation, owing to
its uniquely anionic nature and glycan-chain terminal occupation. Taking
advantage of boronic acid (BA) based SA-recognition chemistry, we here
demonstrate a label-free, no enzymatic, potentiometric determination of
fetuin, a blood-circulating glycoprotein implicated in physiological and
various pathological states. A phenylboronic acid (PBA)
omega-end-functionalized poly(ethylene glycol) (PEG) with an
alpha-tethering unit bearing pendent alkyne groups was “grafted-to”
a gold electrode modified with 11-azide-undecathiol by a
copper-catalyzed azide-alkyne cycloaddition reaction. Using the
electrode, fetuin was potentiometrically detectable with a mu
M-order-sensitivity that is comparable to what is found in
blood-collected specimen. Our finding may have implications for
developing a remarkably economic hemodiagnostic technology with ease of
downsizing and mass production.
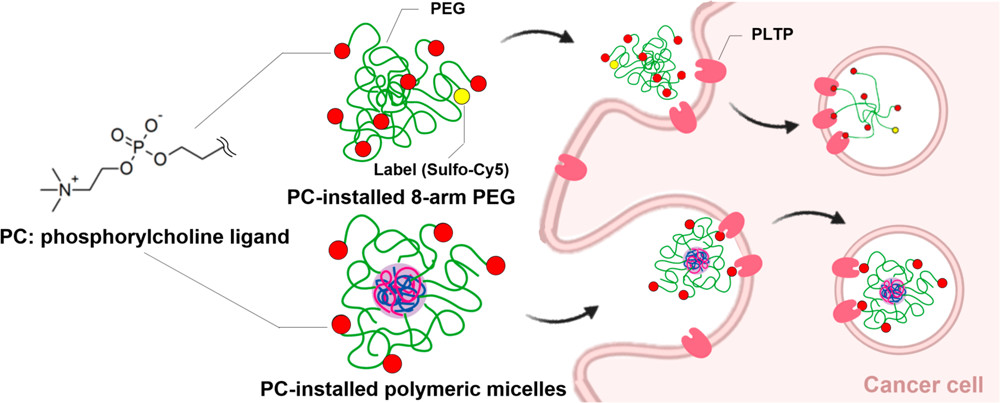 Phosphorylcholine-Installed Nanocarriers Target Pancreatic Cancer Cells
through the Phospholipid Transfer Protein
Phosphorylcholine-Installed Nanocarriers Target Pancreatic Cancer Cells
through the Phospholipid Transfer Protein
Hong, T.; Miyazaki, T.; Matsumoto, A.; Koji, K.; Miyahara, Y.; Anraku, Y.; Cabral, H.
ACS Biomater. Sci. Eng. 2021, 7(9), 4439-4445.
AbstractPhosphorylcholine (PC) has been used to improve the water solubility and biocompatibility of biomaterials. Here, we show that PC can also work as a ligand for targeting cancer cells based on their increased phospholipid metabolism. PC-installed multiarm poly(ethylene glycol)s and polymeric micelles achieved high and rapid internalization in pancreatic cancer cells. This enhanced cellular uptake was drastically reduced when the cells were incubated with excess free PC or at 4 degrees C, as well as by inhibiting the phospholipid transfer protein (PLTP) on the surface of cancer cells, indicating an energy dependent active transport mediated by PLTP.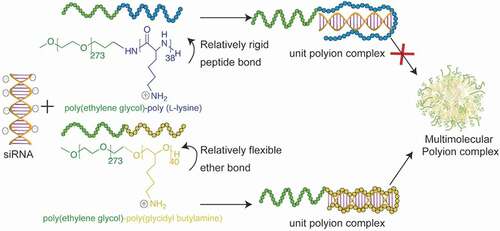 Block catiomer with flexible cationic segment enhances complexation with
siRNA and the delivery performance in vitro
Block catiomer with flexible cationic segment enhances complexation with
siRNA and the delivery performance in vitro
Yang, W.; Miyazaki, T.; Chen, P.; Hong, T.; Naito, M.; Miyahara, Y.; Matsumoto, A.; Kataoka, K.; Miyata, K.; Cabral, H.
Sci. Technol. Adv. Mater. 2021, 22(1), 850-863.
AbstractRNA interference (RNAi) by small interfering RNAs (siRNAs) is a promising therapeutic approach. Because siRNA has limited intracellular access and is rapidly cleared in vivo, the success of RNAi depends on efficient delivery technologies. Particularly, polyion complexation between block catiomers and siRNA is a versatile approach for constructing effective carriers, such as unit polyion complexes (uPIC), core-shell polyion complex (PIC) micelles and vesicular siRNAsomes, by engineering the structure of block catiomers. In this regard, the flexibility of block catiomers could be an important parameter in the formation of PIC nanostructures with siRNA, though its effect remains unknown. Here, we studied the influence of block catiomer flexibility on the assembly of PIC structures with siRNA using a complementary polymeric system, i.e. poly(ethylene glycol)-poly(L-lysine) (PEG-PLL) and PEG-poly(glycidylbutylamine) (PEG-PGBA), which has a relatively more flexible polycation segment than PEG-PLL. Mixing PEG-PGBA with siRNA at molar ratios of primary amines in polymer to phosphates in the siRNA (N/P ratios) higher than 1.5 promoted the multimolecular association of uPICs, whereas PEG-PLL formed uPIC at all N/P ratios higher than 1. Moreover, uPICs from PEG-PGBA were more stable against counter polyanion exchange than uPICs from PEG-PLL, probably due to a favorable complexation process, as suggested by computational studies of siRNA/block catiomer binding. In in vitro experiments, PEG-PGBA uPICs promoted effective intracellular delivery of siRNA and efficient gene knockdown. Our results indicate the significance of polycation flexibility on assembling PIC structures with siRNA, and its potential for developing innovative delivery systems.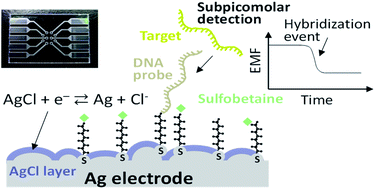 Organic and inorganic mixed phase modification of a silver surface for
functionalization with biomolecules and stabilization of electromotive
force
Organic and inorganic mixed phase modification of a silver surface for
functionalization with biomolecules and stabilization of electromotive
force
Tabata, M.; Kataoka-Hamai, C.; Nogami, K.; Tsuya, D.; Goda, T.; Matsumoto, A.; Miyahara, Y.
RSC Adv. 2021, 11(40), 24958-24967.
AbstractA solid-state potentiometric biosensor based on the organic and inorganic mixed phase modification of a silver surface is proposed. Stabilization of the electromotive force and functionalization with biomolecules on the sensing surface were simultaneously achieved using silver chloride chemically deposited with 1,3-diaminopropanetetraacetic acid ferric ammonium salt monohydrate and a self-assembled monolayer with oligonucleotide probes, respectively. The formation of silver chloride and adsorption of alkanethiol on the silver surface were confirmed with X-ray photoelectron spectroscopy. The resulting modified surface reduced the nonspecific binding of interfering biomolecules and achieved a high signal to noise ratio. The electromotive forces of the modified silver thin film electrodes were stable under constant chloride ion concentrations. Hybridization assays were performed to detect microRNA 146. The lower limit of detection was 0.1 pM because of the small standard deviation. The proposed biosensor could be useful as a disposable single-use sensor in medical fields such as liquid biopsies.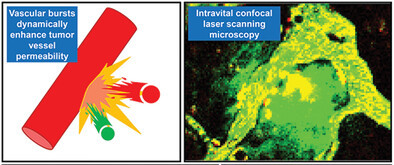 Vascular Bursts Act as a Versatile Tumor Vessel Permeation Route for
Blood-Borne Particles and Cells
Vascular Bursts Act as a Versatile Tumor Vessel Permeation Route for
Blood-Borne Particles and Cells
Igarashi, K.; Cabral, H.; Hong, T.; Anraku, Y.; Mpekris, F.; Stylianopoulos, T.; Khan, T.; Matsumoto, A.; Kataoka, K.; Matsumoto, Y.; Yamasoba, T.
Small 2021, 17(42), 2103751.
AbstractThe recent discovery of vascular bursts in tumor vasculature has sparked renewed interest in active transport of sub-100 nm particles, highlighting the dynamic nature of its distribution. In this paper, an even broader versatility of vascular burst transport is demonstrated through the validation of a library of blood-borne particles together with mathematical model simulation to estimate the burst vent size.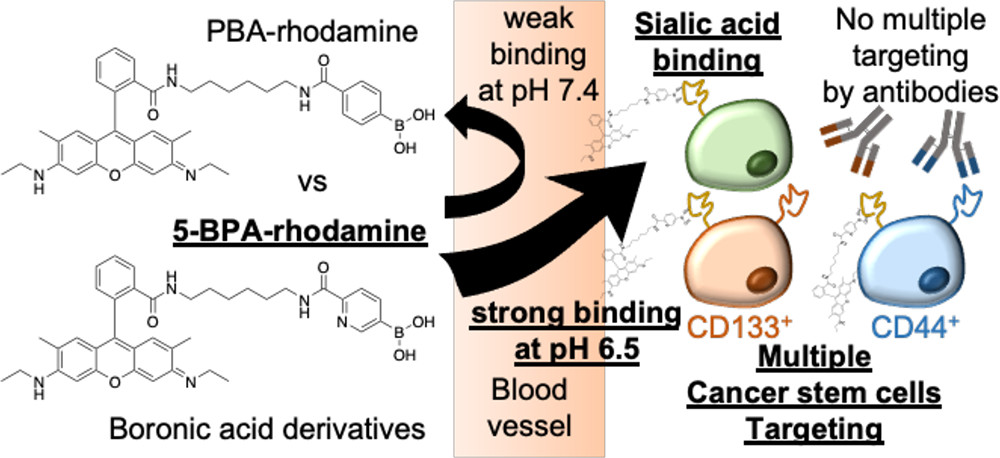 Boronic Acid Ligands Can Target Multiple Subpopulations of Pancreatic Cancer Stem Cells via pH-Dependent Glycan-Terminal Sialic Acid Recognition
Boronic Acid Ligands Can Target Multiple Subpopulations of Pancreatic Cancer Stem Cells via pH-Dependent Glycan-Terminal Sialic Acid Recognition
Miyazaki, T.; Khan, T.; Tachihara, Y.; Itoh, M.; Miyazawa, T.; Suganami, T.; Miyahara, Y.; Cabral, H.; Matsumoto, A.
ACS Appl. Bio Mater. 2021, 4(9), 6647-6651.
AbstractEradication of cancer stem cells (CSCs) is an ultimate goal in cancer chemotherapy. Although a ligand-assisted targeting approach seems rational, the existence of subpopulations of CSCs and their discrimination from those present on healthy sites makes it a severe challenge. Some boronic acid (BA) derivatives are known for the ability to bind with glycan-terminal sialic acid (SA), in a manner dependent on the acidification found in hypoxic tumoral microenvironment. Taking advantage of this feature, here we show that the BA–ligand fluorescence conjugate can effectively target multiple CSC subpopulations in parallel, which otherwise must be independently aimed when using antibody-ligands.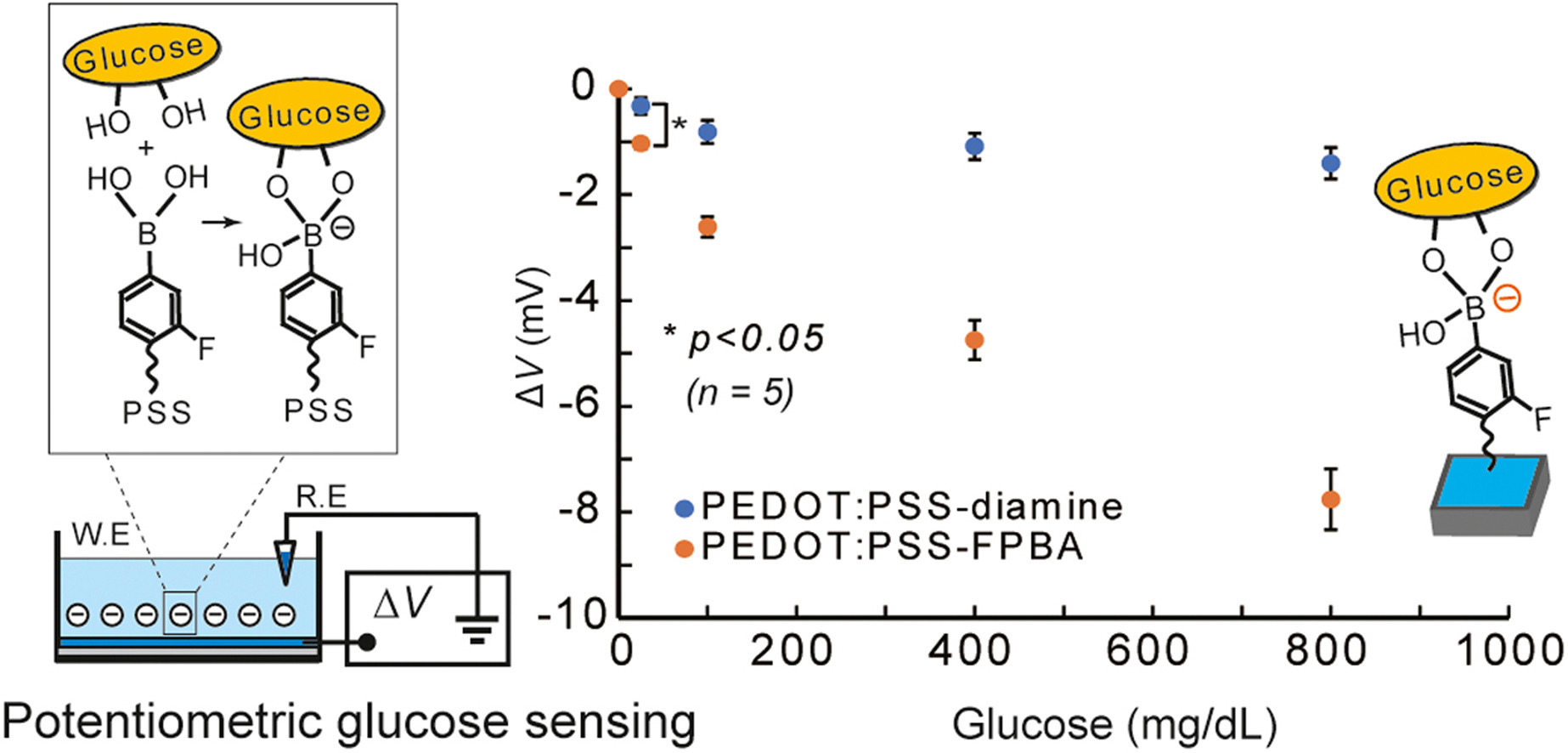 In-situ chemical modification of printed conducting polymer films
for specific glucose biosensing
In-situ chemical modification of printed conducting polymer films
for specific glucose biosensing
Fujisaki, H.; Watcharawittayakul, T.; Matsumoto, A.; Miyahara, Y.; Goda, T.
Sens. Actuators B: Chem. 2021, 349.
AbstractConductive polymers, such as poly(3,4-ethylenedioxythiophene):poly(styrene sulfonate) (PEDOT:PSS), can be used for specific biosensing after its surface is modified with functional groups. Herein, we report a simple two-step surface modification method for commercially available PEDOT:PSS films printed on a flexible substrate. The derivatization process involves: (1) the introduction of an ethylenediamine linker for conjugation with the biorecognition element by activating the sulfonate group in PSS with an acid chloride reaction; and (2) the introduction of FPBA into the ethylenediamine linker. As a proof of concept, 4-carboxy-3-fluorophenylboronic acid (carboxy FPBA) is covalently introduced to the side chain of the PSS dopant in the conductive layer. FPBA serves as a selective molecular recognition element for diol compounds, including glucose, at neutral pH. Continuous glucose monitoring is achieved by label-free potentiometry using FPBA-functionalized commercial PEDOT:PSS films. Negative potential responses with glucose are successfully achieved in ideal and realistic buffer solutions with or without 1% fetal bovine serum (FBS) because of the complexation of FPBA. The dynamic range of the enzyme-free sensor covered physiologically relevant blood glucose levels of 70-140 mg/dL. The developed method is suitable and cost effective for the mass production of biosensors as it can be applied to a large surface area at once. Furthermore, various ligands and receptors can be introduced onto commercial PEDOT:PSS films using this technique. Hence, the proposed method has potential applications in wearable and implantable biosensors.
Reviews
ボロン酸ゲルを用いた糖尿病治療デバイスの社会実装 (Social implementation of diabetes treatment device using boronic acid gel)
松元 亮; 宮崎 拓也; 伊藤 美智子; 菅波 孝祥; 宮原 裕二.
バイオマテリアル (Journal of Japanese Society for Biomaterials) 2021, 39(4), 252-257.
A boronate gel-based synthetic platform for closed-loop insulin delivery systems
Matsumoto, A.; Chen, S.
Polym. J. 2021, 53(12), 1305-1314.
AbstractDiabetes is one of the most devastating global diseases with an ever-increasing number of patients. Achieving persistent glycemic control in a painless and convenient way is an unmet goal for diabetes management. Insulin therapy is commonly utilized for diabetes treatment and usually relies on patient self-injection. This not only impairs a patient’s quality of life and fails to precisely control the blood glucose level but also brings the risk of life-threatening hypoglycemia. “closed-loop” insulin delivery systems could avoid these issues by providing on-demand insulin delivery. However, safety concerns limit the application of currently developed electronics-derived or enzyme-based systems. Phenylboronic acid (PBA), with the ability to reversibly bind glucose and a chemically tailored binding specificity, has attracted substantial attention in recent years. This focus review provides an overview of PBA-based versatile insulin delivery platforms developed in our group, including new PBA derivatives, glucose-responsive gels, and gel-combined medical devices, with a unique “skin layer” controlled diffusion feature. Boronic acids (BA) are known for their ability to reversibly interact with the diol groups, a common motif of biomolecules including sugars, ribose, and catechols. One can tailor BAs to elicit a divergent profile of binding strength and specificity on the basis of stereochemistry and controlled electronic effects. This focus review provides an overview of a phenylboronic acid (PBA)-based, totally synthetic platform for insulin delivery applications developed in our group, with focuses on the development of new PBA derivatives, glucose-responsive gels, and the gel-combined medical devices.マクロファージにおける脂質代謝障害に着目したNASH発症機構の解明 (Elucidation of the mechanism of NASH onset focusing on lipid metabolic disorders in macrophages)
伊藤 美智子; 金井 紗綾香; 田村 篤志; 白川 伊吹; 金森 耀平; 田中 都; 松元 亮; 宮原 裕二; 小川 佳宏; 菅波 孝祥.
日本内分泌学会雑誌 (Journal of the Japanese Endocrine Society) 2021, 97(1), 282.
2020
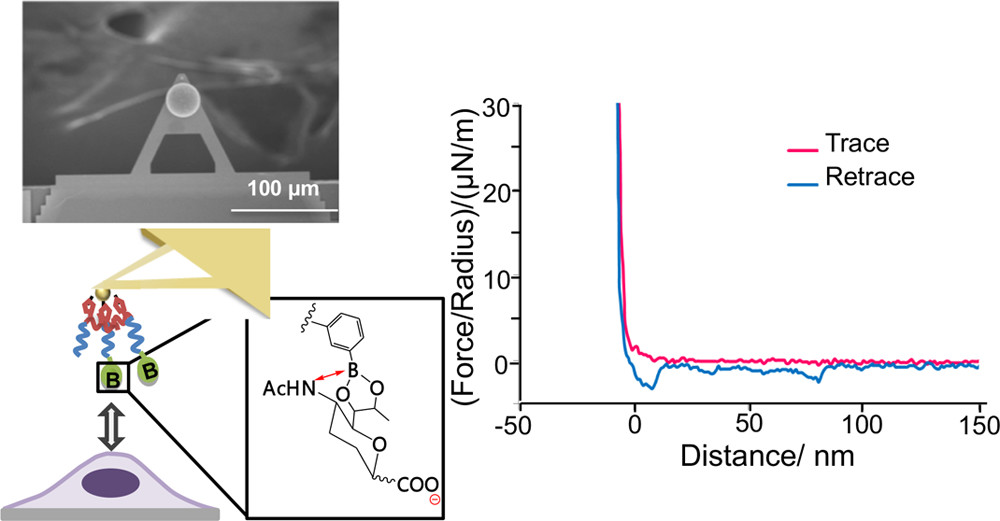 Direct Observation of Cell Surface Sialylation by Atomic Force
Microscopy Employing Boronic Acid-Sialic Acid Reversible Interaction
Direct Observation of Cell Surface Sialylation by Atomic Force
Microscopy Employing Boronic Acid-Sialic Acid Reversible Interaction
Osawa, S.; Matsumoto, A.; Maejima, Y.; Suzuki, T.; Miyahara, Y.; Otsuka, H.
Anal. Chem. 2020, 92(17), 11714-11720.
AbstractTracing cell surface sialylation dynamics at a scale of the glycolipoprotein microdomain (lipid rafts) formations remains an intriguing challenge of cellular biology. Here, we demonstrate that this goal is accessible, taking advantage of a boronic acid (BA)-based reversible molecular recognition chemistry. A BA-end-functionalized poly(ethylene glycol) was decorated onto an atomic force microscopy (AFM) cantilever, which provided a dynamic and sialic acid (SA)-specific imaging mode. Using this technique, we were able to heat map the SA expression levels not only on protein-decorated substrates but also directly on the cell surfaces, with a submicrometer scale resolution that may be relevant to that of the lipid rafts formation. The SA specificity and the binding reversibility of the probe were confirmed from its pH-dependent characteristics and an inhibition assay using free state SA. This finding may provide a noninvasive means for assessing a variety of SA-involved glycosylation dynamics spanning from physiology to pathology.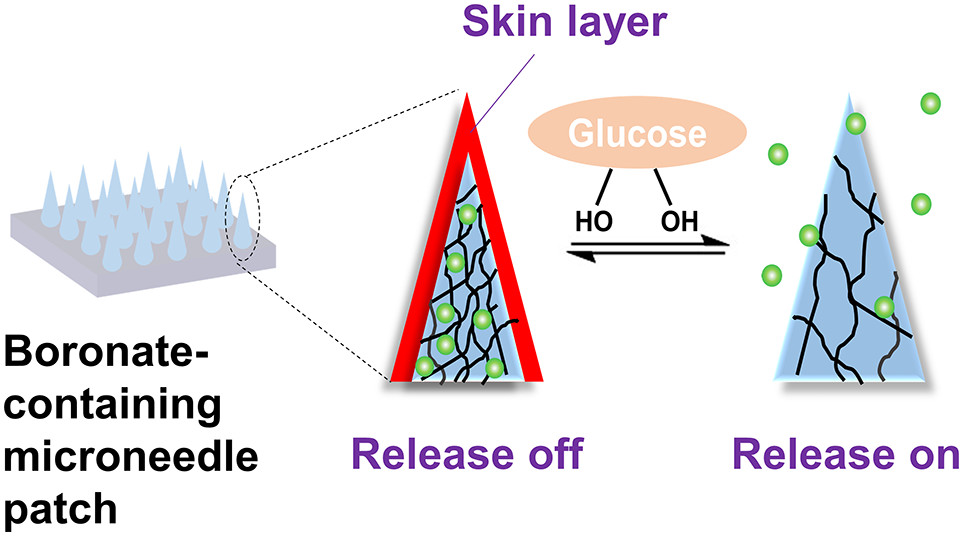 Temperature-Stable Boronate Gel-Based Microneedle Technology for
Self-Regulated Insulin Delivery
Temperature-Stable Boronate Gel-Based Microneedle Technology for
Self-Regulated Insulin Delivery
Chen, S.; Miyazaki, T.; Itoh, M.; Matsumoto, H.; Moro-oka, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.; Matsumoto, A.
ACS Appl. Polym. Mater. 2020, 2(7), 2781-2790.
Abstract/Cover Insulin delivery in a self-regulated and painless way to tightly control
the glycemic level is highly demanded for diabetes treatment.
Phenylboronic acid (PBA) has gained great research interests due to its
synthetic nature and reversible binding capability with glucose. A
totally synthetic smart PBA hydrogel exhibiting efficient glucose
sensitivity at physiological pH and temperature has been previously
developed. However, its clinical applications may be hampered by the
temperature-dependent release profile. Herein, we report a
glucose-responsive, temperature-stable, boronate-containing hydrogel
with optimized formulation and its fabrication into a microneedle (MN)
patch to provide on-demand and convenient insulin delivery. The
resulting MN patch displayed temperature-independent and
glucose-responsive insulin release in a rapid and sustained manner
through the regulation by the “skin layer” formed on the surface.
This MNs patch can effectively penetrate the skin and was highly
biocompatible. Compared to the majority of the glucose-responsive MN
patches capitalizing on glucose oxidase and nanoparticles, this totally
synthetic, protein-free, and nanoparticle-free MN patch could eliminate
the safety concerns and provide the sustainability and advantage for
large-scale production.
Insulin delivery in a self-regulated and painless way to tightly control
the glycemic level is highly demanded for diabetes treatment.
Phenylboronic acid (PBA) has gained great research interests due to its
synthetic nature and reversible binding capability with glucose. A
totally synthetic smart PBA hydrogel exhibiting efficient glucose
sensitivity at physiological pH and temperature has been previously
developed. However, its clinical applications may be hampered by the
temperature-dependent release profile. Herein, we report a
glucose-responsive, temperature-stable, boronate-containing hydrogel
with optimized formulation and its fabrication into a microneedle (MN)
patch to provide on-demand and convenient insulin delivery. The
resulting MN patch displayed temperature-independent and
glucose-responsive insulin release in a rapid and sustained manner
through the regulation by the “skin layer” formed on the surface.
This MNs patch can effectively penetrate the skin and was highly
biocompatible. Compared to the majority of the glucose-responsive MN
patches capitalizing on glucose oxidase and nanoparticles, this totally
synthetic, protein-free, and nanoparticle-free MN patch could eliminate
the safety concerns and provide the sustainability and advantage for
large-scale production.
Hollow fiber-combined glucose-responsive gel technology as an in vivo electronics-free insulin delivery system
Matsumoto, A.; Kuwata, H.; Kimura, S.; Matsumoto, H.; Ochi, K.; Moro-oka, Y.; Watanabe, A.; Yamada, H.; Ishii, H.; Miyazawa, T.; Chen, S.; Baba, T.; Yoshida, H.; Nakamura, T.; Inoue, H.; Ogawa, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.
Commun. Biol. 2020, 3(313).
Abstract Press releaseAccumulating evidence demonstrates that not only sustained elevation of blood glucose levels but also the glucose fluctuation represents key determinants for diabetic complications and mortality. Current closed-loop insulin therapy option is limited to the use of electronics-based systems, although it poses some technical issues with high cost. Here we demonstrate an electronics-free, synthetic boronate gel-based insulin-diffusion-control device technology that can cope with glucose fluctuations and potentially address the electronics-derived issues. The gel was combined with hemodialysis hollow fibers and scaled suitable for rats, serving as a subcutaneously implantable, insulin-diffusion-active site in a manner dependent on the subcutaneous glucose. Continuous glucose monitoring tests revealed that our device not only normalizes average glucose level of rats, but also markedly ameliorates the fluctuations over timescale of a day without inducing hypoglycemia. With inherent stability, diffusion-dependent scalability, and week-long & acute glucose-responsiveness, our technology may offer a low-cost alternative to current electronics-based approaches. Here, the authors develop an in vivo insulin delivery system which consists of a glucose responsive gel combined with hemodialysis hollow fibers. This system is electronics-free, temperature independent, and can stably sustain acute glucose-responsiveness in rats.C-type lectin Mincle mediates cell death-triggered inflammation in acute kidney injury
Tanaka, M.; Saka-Tanaka, M.; Ochi, K.; Fujieda, K.; Sugiura, Y.; Miyamoto, T.; Kohda, H.; Ito, A.; Miyazawa, T.; Matsumoto, A.; Aoe, S.; Miyamoto, Y.; Tsuboi, N.; Maruyama, S.; Suematsu, M.; Yamasaki, S.; Ogawa, Y.; Suganami, T.
J. Exp. Med. 2020, 217(11), e20192230.
AbstractAccumulating evidence indicates that cell death triggers sterile inflammation and that impaired clearance of dead cells causes nonresolving inflammation; however, the underlying mechanisms are still unclear. Here, we show that macrophage-inducible C-type lectin (Mincle) senses renal tubular cell death to induce sustained inflammation after acute kidney injury in mice. Mincle-deficient mice were protected against tissue damage and subsequent atrophy of the kidney after ischemia-reperfusion injury. Using lipophilic extract from the injured kidney, we identified beta-glucosylceramide as an endogenous Mincle ligand. Notably, free cholesterol markedly enhanced the agonistic effect of beta-glucosylceramide on Mincle. Moreover, beta-glucosylceramide and free cholesterol accumulated in dead renal tubules in proximity to Mincle-expressing macrophages, where Mincle was supposed to inhibit clearance of dead cells and increase proinflammatory cytokine production. This study demonstrates that beta-glucosylceramide in combination with free cholesterol acts on Mincle as an endogenous ligand to induce cell death-triggered, sustained inflammation after acute kidney injury.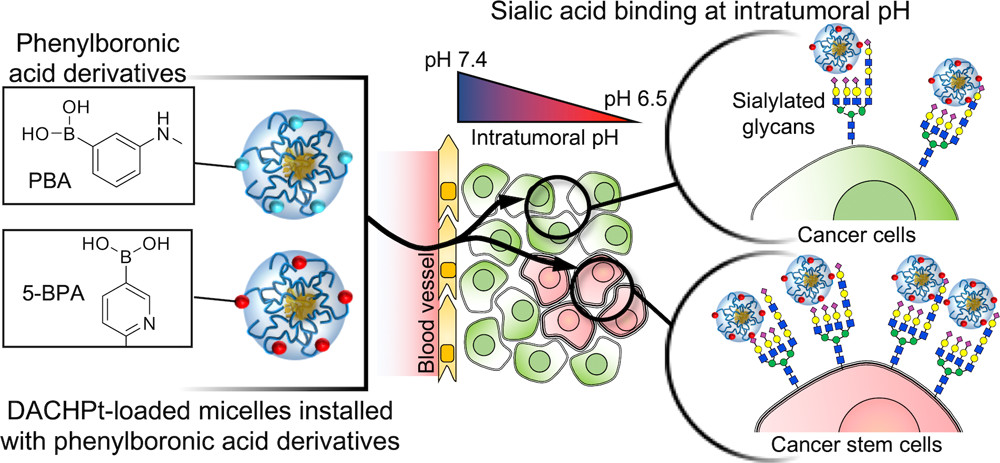 Structural Control of Boronic Acid Ligands Enhances Intratumoral Targeting of Sialic Acid To Eradicate Cancer Stem-like Cells
Structural Control of Boronic Acid Ligands Enhances Intratumoral Targeting of Sialic Acid To Eradicate Cancer Stem-like Cells
Khan, T.; Igarashi, K.; Tanabe, A.; Miyazawa, T.; Fukushima, S.; Miura, Y.; Matsumoto, Y.; Yamasoba, T.; Matsumoto, A.; Cabral, H.; Kataoka, K.
ACS Appl. Bio Mater. 2020, 3(8), 5030-5039.
AbstractAberrant sialylation of cancer cells is emerging as an attractive method for generating effective antitumor strategies. However, as sialic acid (SA) is also present in healthy tissues, systems targeting SA in tumors must be strategically designed to be specifically activated in an intratumoral environment while avoiding systemic interaction. Phenylboronic acid (PBA) and its derivatives have shown potential for developing such smart ligands based on its triggered binding to SA at intratumoral pH. Because the affinity of PBAs against SA can be structurally controlled, the approach may further offer the possibility to enhance tumor targeting by molecularly engineering PBAs. Thus, to demonstrate that the modification of the chemical structure of PBAs can promote tumor targeting, we compared nanomedicines installed with the standard PBA or 5-boronopicolinic acid (5-BPA), which shows an exceptionally high binding affinity to SA in acidic pH. Platinum anticancer drugs were loaded into these nanomedicines and evaluated against orthotopic head and neck tumors, featuring a large fraction of SA-rich cancer stem-like cells (CSCs) that are resistant to platinum drugs. The 5-BPA ligands increased intracellular drug delivery of nanomedicines at intratumoral pH (pH 6.5) and enhanced the accumulation of nanomedicines in tumors to efficaciously eliminate the malignant CSCs, suppress tumor growth, and prolong mice survival. These findings indicate the potential of engineered PBA ligands for developing effective strategies targeting SA in tumors.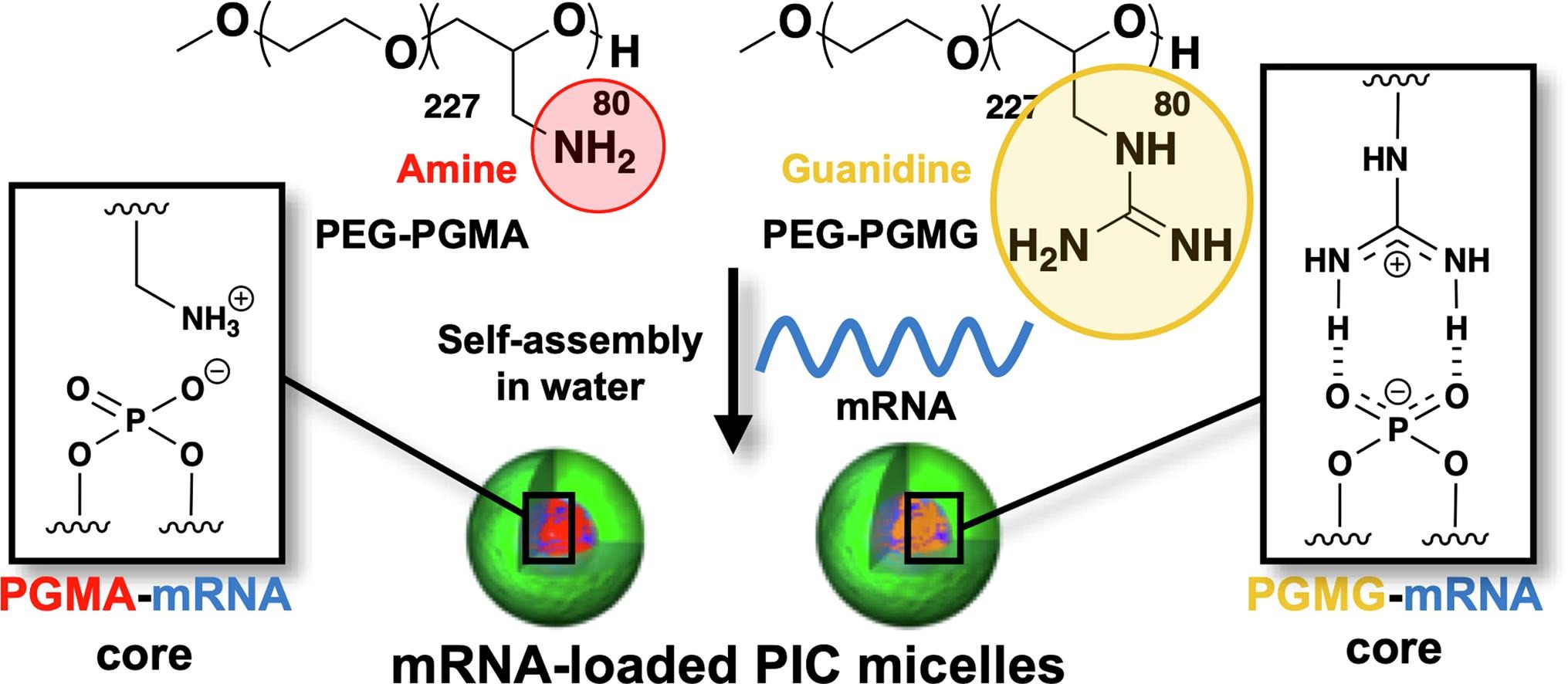 Guanidine-phosphate interactions stabilize polyion complex micelles
based on flexible catiomers to improve mRNA delivery
Guanidine-phosphate interactions stabilize polyion complex micelles
based on flexible catiomers to improve mRNA delivery
Miyazaki, T.; Uchida, S.; Hatano, H.; Miyahara, Y.; Matsumoto, A.; Cabral, H.
Eur. Polym. J. 2020, 140, 110028.
AbstractMessenger RNA (mRNA) holds great potential as therapeutic agent, though its broad applicability is hampered by its rapid degradation by nuclease attacks and poor access to cells. Polyion complex (PIC) micelles comprising block catiomers are promising mRNA carriers, because of their effective mRNA protection in the PIC core and the capability to promote mRNA cellular uptake. We have recently shown that flexible polycations can promote the interaction between catiomers and mRNA toward improving delivery efficiency, demonstrating that, by engineering the PIC formation, the efficacy of mRNA-loaded PIC micelles (mRNA/m) can be improved. Herein, we focused on the valency between the polycation segment and mRNA, and studied its ability to promote, in collaboration with the flexible catiomers, mRNA stability and delivery efficiency for producing potent nanocarriers. The functionalities of guanidine and primary amine were compared through preparation of two different block copolymers, namely, poly(ethylene glycol)-poly(glycidyl methyl guanidine) (PEG-PGMG) and PEG-poly(glycidyl methyl amine (PEG-PGMA)). mRNA/m from PEG-PGMG (mRNA/mPGMG) showed enhanced stability against biological molecules, including polyanions, urea and nucleases compared to mRNA/m prepared from PEG-PGMA (mRNA/mPGMA), resulting in high efficiency of cellular uptake and protein translation. Our results indicate that multivalent interactions between the guanidine moieties in the polycation segments of block copolymers and the phosphate groups in mRNA are a key factor for stabilizing mRNA/m toward improving mRNA delivery efficiency.
2019
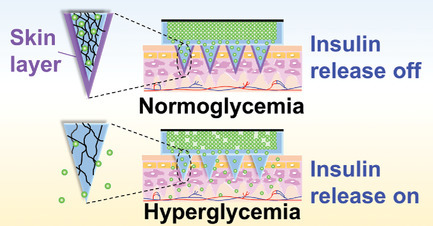 Microneedle-Array Patch Fabricated with Enzyme-Free Polymeric Components Capable of On-Demand Insulin Delivery
Microneedle-Array Patch Fabricated with Enzyme-Free Polymeric Components Capable of On-Demand Insulin Delivery
Chen, S.; Matsumoto, H.; Moro-oka, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.; Matsumoto, A.
Adv. Funct. Mater. 2019, 29(7).
Abstract/Cover Video abstract Press release Achieving persistent glycemic control in a painless and convenient way
is the ultimate goal of diabetes management. Herein, an enzyme-free
polymeric microneedle (MN)-array patch composed of a boronate-containing
hydrogel semi-interpenetrated by biocompatible silk fibroin is
developed. Consistent with the previous reports, the presence of the
boronate-hydrogel allows for glucose-responsive diffusion-control of
insulin, while the crystalline fibroin component serves as a
matrix-stiffener to validate skin penetration. Remarkably, this
enzyme-free smart artificial on-skin pancreas prototype remains stable
for at least 2 months in an aqueous environment. Furthermore, it
establishes sustained as well as acute glucose-responsive insulin
delivery, and is to the authors’ knowledge, the first successful
material design addressing such two technical challenges at once on an
MN format. This long-acting, on-demand insulin delivery technology may
offer a candidate for a next-generation diabetes therapy that is
remarkably stable, safe, economically efficient, and capable of
providing both acute- and continuous glycemic control in a manner
minimally dependent on patient compliance.
Newspaper report
Achieving persistent glycemic control in a painless and convenient way
is the ultimate goal of diabetes management. Herein, an enzyme-free
polymeric microneedle (MN)-array patch composed of a boronate-containing
hydrogel semi-interpenetrated by biocompatible silk fibroin is
developed. Consistent with the previous reports, the presence of the
boronate-hydrogel allows for glucose-responsive diffusion-control of
insulin, while the crystalline fibroin component serves as a
matrix-stiffener to validate skin penetration. Remarkably, this
enzyme-free smart artificial on-skin pancreas prototype remains stable
for at least 2 months in an aqueous environment. Furthermore, it
establishes sustained as well as acute glucose-responsive insulin
delivery, and is to the authors’ knowledge, the first successful
material design addressing such two technical challenges at once on an
MN format. This long-acting, on-demand insulin delivery technology may
offer a candidate for a next-generation diabetes therapy that is
remarkably stable, safe, economically efficient, and capable of
providing both acute- and continuous glycemic control in a manner
minimally dependent on patient compliance.
Newspaper report- 熊本日日新聞「「貼るだけ医薬品」開発進む」 (2019年3月6日, p.12)
- 京都新聞「進む「貼るだけ医薬品」」 (2019年3月6日夕刊, p.2)
- 山陽新聞「開発進む「貼るだけ医薬品」」 (2019年3月3日, p.25)
- 四国新聞「手軽で患者負担少なく 進む「貼るだけ医薬品」開発」 (2019年3月1日, p.13)
- 福井新聞「貼る医薬品開発進む」(2019年2月26日, p.14)
- 山陰中央新聞「ワクチン, 人工すい臓も―「貼るだけ医薬品」開発」 (2019年2月25日, p.6)
- 大分合同新聞「貼る医薬品 開発進む」 (2019年2月25日, p.4)
- 下野新聞「「薬や注射の手間なし―「貼るだけ医薬品」進化」 (2019年2月22日, p.21)
- 北海道新聞「貼るだけ医薬品 進む開発」 (2019年2月20日夕刊, p.4)
- 琉球新聞「進む「貼るだけ医薬品」」(2019年2月20日, p.29)
- 山形新聞「体にぺたり 医療を変える」(2019年2月20日, p.14)
- 徳島新聞「開発進む「貼るだけ医薬品」」 (2019年2月18日夕刊, p.2)
- 中国新聞「貼る医薬品 進む開発」 (2019年2月17日, p.11)
- 高知新聞「進む「貼るだけ医薬品」」 (2019年2月14日夕刊, p.4)
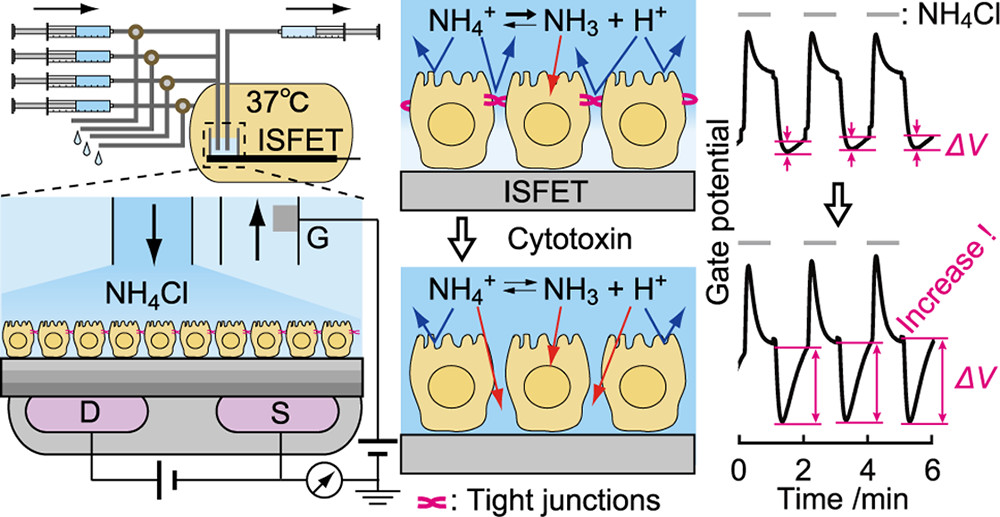 Induced Proton Perturbation for Sensitive and Selective Detection of
Tight Junction Breakdown
Induced Proton Perturbation for Sensitive and Selective Detection of
Tight Junction Breakdown
Hatano, H.; Goda, T.; Matsumoto, A.; Miyahara, Y.
Anal. Chem. 2019, 91(5), 3525-3532.
Abstract/Cover Tight junctions (TJs) in the epithelial cell gap play primary roles in
body defense and nutrient absorption in multicellular organisms.
Standard in vitro assays lack sensitivity, selectivity, temporal
resolution, and throughput for bioengineering applications. Prompted by
the rigorous barrier functions of TJ, we developed a TJ assay that
senses proton leaks in the cell gap using ion-sensitive field-effect
transistors. Upon exposure of Madin-Darby canine kidney (MDCK) cells
cultured on a gate dielectric to a calcium-chelator EGTA,
ammonia-assisted pH perturbation was enhanced solely in TJ-forming
cells. This was supported by simulations with increased ion permeability
in the paracellular pathway. Following administration of Clostridium
perfringens enterotoxin as a specific TJ inhibitor to the MDCK cells,
our method detected TJ breakdown at a 13X lower concentration than a
conventional trans-epithelial electrical resistance assay. Thus, the
semiconductor-based active pH sensing could offer an alternative to
current in vitro assays for TJs in pathological, toxicological, and
pharmaceutical studies.
Tight junctions (TJs) in the epithelial cell gap play primary roles in
body defense and nutrient absorption in multicellular organisms.
Standard in vitro assays lack sensitivity, selectivity, temporal
resolution, and throughput for bioengineering applications. Prompted by
the rigorous barrier functions of TJ, we developed a TJ assay that
senses proton leaks in the cell gap using ion-sensitive field-effect
transistors. Upon exposure of Madin-Darby canine kidney (MDCK) cells
cultured on a gate dielectric to a calcium-chelator EGTA,
ammonia-assisted pH perturbation was enhanced solely in TJ-forming
cells. This was supported by simulations with increased ion permeability
in the paracellular pathway. Following administration of Clostridium
perfringens enterotoxin as a specific TJ inhibitor to the MDCK cells,
our method detected TJ breakdown at a 13X lower concentration than a
conventional trans-epithelial electrical resistance assay. Thus, the
semiconductor-based active pH sensing could offer an alternative to
current in vitro assays for TJs in pathological, toxicological, and
pharmaceutical studies.
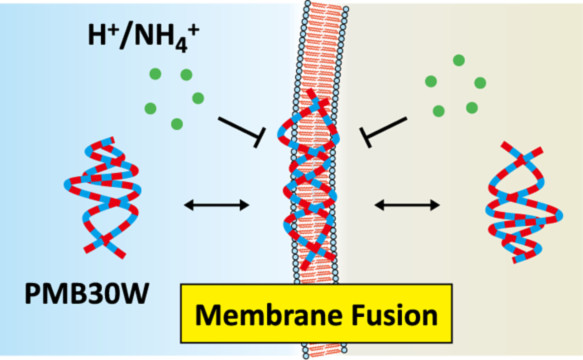 Translocation Mechanisms of Cell-Penetrating Polymers Identified by
Induced Proton Dynamics
Translocation Mechanisms of Cell-Penetrating Polymers Identified by
Induced Proton Dynamics
Goda, T.; Imaizumi, Y.; Hatano, H.; Matsumoto, A.; Ishihara, K.; Miyahara, Y.
Langmuir 2019, 35(24), 8167-8173.
AbstractUnlike the majority of nanomaterials designed for cellular uptake via endocytic pathways, some of the functional nanoparticles and nanospheres directly enter the cytoplasm without overt biomembrane injuries. Previously, we have shown that a water-soluble nanoaggregate composed of amphiphilic random copolymer of 2-methacryloyloxyethyl phosphorylcholine (MPC) and n-butyl methacrylate (BMA), poly(MPC-random-BMA) (PMB), passes live cell membranes in an endocytosis-free manner. Yet, details in its translocation mechanism remain elusive due to the lack of proper analytical methods. To understand this phenomenon experimentally, we elaborated the original pH perturbation assay that is extremely sensitive to the pore formation on cell membranes. The ultimate sensitivity originates from the detection of the smallest indicator H+ (H3O+) passed through the molecularly sized transmembrane pores upon challenge by exogenous reagents. We revealed that water-soluble PMB at the 30 mol % MPC unit (i.e., PMB30W) penetrated into the cytosol of model mammalian cells without any proton leaks, in contrast to conventional cell-penetrating peptides, TAT and R8 as well as the surfactant, Triton X-100. While exposure of PMB30W permeabilized cytoplasmic lactate dehydrogenase out of the cells, indicating the alteration of cell membrane polarity by partitioning of amphiphilic PMB30W into the lipid bilayers. Nevertheless, the biomembrane alterations by PMB30W did not exhibit cytotoxicity. In summary, elucidating translocation mechanisms by proton dynamics will guide the design of nanomaterials with controlled permeabilization to cell membranes for bioengineering applications.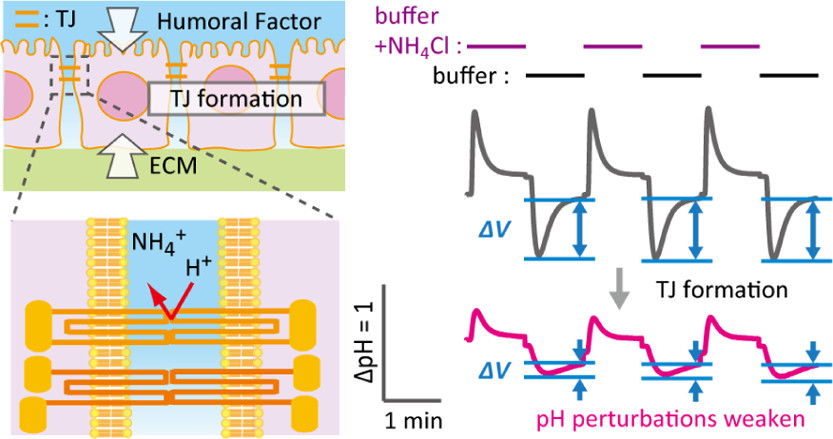 Induced Proton Dynamics on Semiconductor Surfaces for Sensing Tight
Junction Formation Enhanced by an Extracellular Matrix and Drug
Induced Proton Dynamics on Semiconductor Surfaces for Sensing Tight
Junction Formation Enhanced by an Extracellular Matrix and Drug
Hatano, H.; Goda, T.; Matsumoto, A.; Miyahara, Y.
ACS Sens. 2019, 4(12), 3195-3202.
AbstractIn the fields of tissue engineering and drug discovery, confirming the formation and maturation of epithelial cell tight junctions (TJs), which are necessary for blocking pathogenic invasion and absorption of nutrients and ions, at a high spatiotemporal resolution is essential. We previously developed a system of monitoring pH perturbation induced by weak acid exposure to cells cultured on an ion-sensitive field-effect transistor that enables a sensitive and specific detection of biomembrane injuries and TJ breakdowns caused by external stimuli such as nanomaterials and cytotoxins. In this study, we monitor time-lapse changes in the paracellular diffusion of growing epithelial cell monolayers using the pH perturbation assay as well as conventional permeability and trans-epithelial electrical resistance assays. The effects of the extracellular matrix and a TJ potentiator (KN-93) on epithelial TJ formation are evaluated. TJ formations were promoted on the substrate coated with Matrigel more than on the one coated with poly(L-lysine). KN-93 accelerated TJ formations in a dose-dependent manner. The pH perturbation assay denoted a longer incubation time for the completion of TJ formation compared with the conventional assays under the same conditions. Importantly, the pH perturbation assay is able to rigorously evaluate TJ formation, as the assay uses protons as the smallest indicator for detecting paracellular gaps, and the pH perturbation is specific to TJ alterations. These features for in vitro TJ evaluation using proton dynamics are advantageous for applications in tissue engineering and drug development.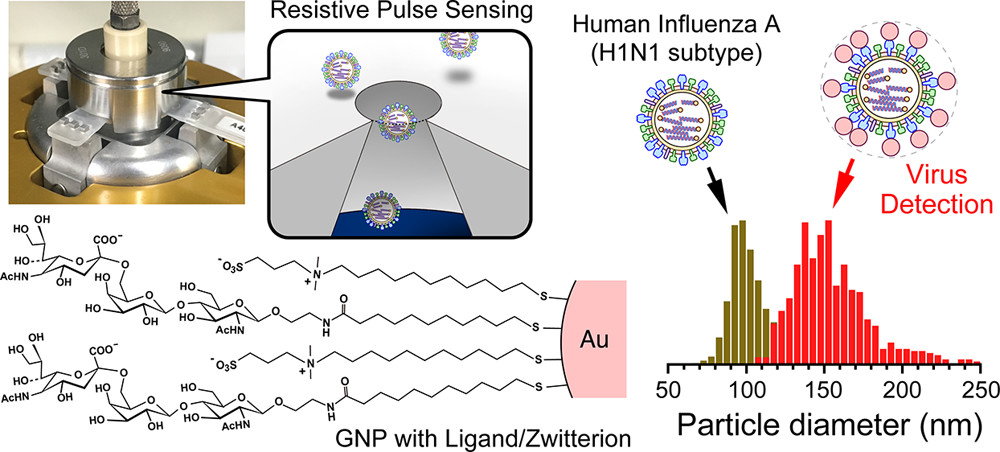 Gold Nanoparticles with Ligand/Zwitterion Hybrid Layer for Individual
Counting of Influenza A H1N1 Subtype Using Resistive Pulse Sensing
Gold Nanoparticles with Ligand/Zwitterion Hybrid Layer for Individual
Counting of Influenza A H1N1 Subtype Using Resistive Pulse Sensing
Horiguchi, Y.; Goda, T.; Matsumoto, A.; Takeuchi, H.; Yamaoka, S.; Miyahara, Y.
Langmuir 2019, 35(5), 1798-1806.
AbstractResistive pulse sensing (RPS) is an analytical technique for detecting particles with nano- to micrometer diameters, such as proteins, viruses, and bacteria. RPS is a promising tool for diagnosis as it can analyze the characteristics of target particles individually from ion current blockades as pulse waveforms. However, it is difficult to discriminate analog targets because RPS merely provides physical information such as size, shape, concentration, and charge density of the analyte. Influenza A virus, which is 80-120 nm in diameter, has various subtypes, demonstrating the diversity of virus characteristics. For example, highly pathogenic avian influenza infections in humans are recognized as an emerging infectious disease with high mortality rates compared with human influenza viruses. Distinguishing human from avian influenza using their differing biological characteristics would be challenging using RPS. To develop a highly selective diagnostic system for infectious diseases, we combined RPS with molecular recognition. Gold nanoparticles (GNPs) that have human influenza A (H1N1 subtype) virus-specific sialic acid receptors on the surface were prepared as a virus label for RPS analysis. A sulfobetaine and sialic acid (ligand) hybrid surface was formed on the GNPs for the suppression of nonspecific interaction. The results show a size change of viruses derived from specific interactions with GNPs. In contrast, no size shift was observed when nonspecific sialic acid receptor immobilized GNPs were used. Detection of viruses by individual particle counting could be a new facet of diagnosis.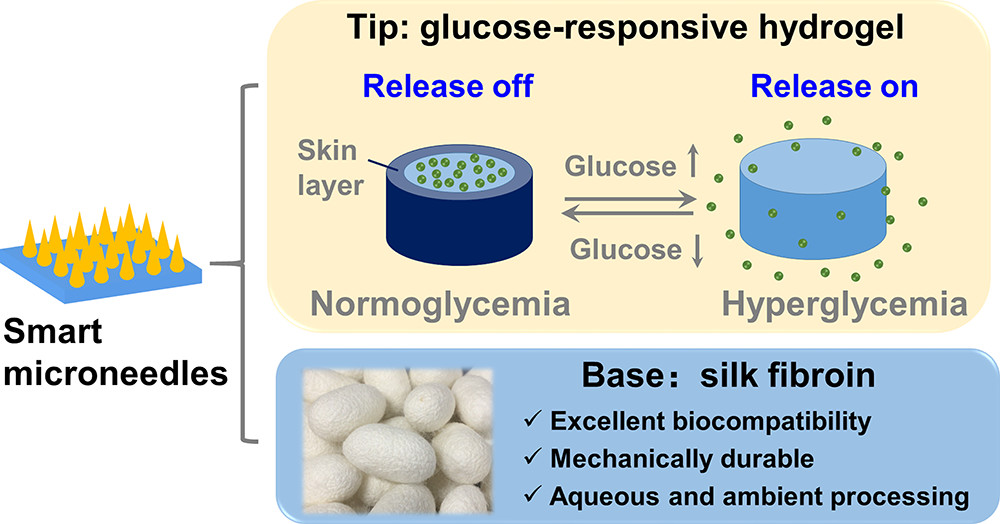 Smart Microneedle Fabricated with Silk Fibroin Combined Semi
interpenetrating Network Hydrogel for Glucose-Responsive Insulin
Delivery
Smart Microneedle Fabricated with Silk Fibroin Combined Semi
interpenetrating Network Hydrogel for Glucose-Responsive Insulin
Delivery
Chen, S.; Matsumoto, H.; Moro-oka, Y.; Tanaka, M.; Miyahara, Y.; Suganami, T.; Matsumoto, A.
ACS Biomater. Sci. Eng. 2019, 5(11), 5781-5789.
AbstractMicroneedle (MN) technology, which can transdermally deliver insulin in a noninvasive manner, offers a promising way to replace subcutaneous self-injection for diabetes management. Hydrogel is an attractive candidate for MN fabrication because of its biocompatibility, controllable degradability, and possibility to achieve sustained as well as stimuli-responsive drug delivery. Herein, we report a smart MN composed of a semi-interpenetrating network (semi-IPN) hydrogel prepared by biocompatible silk fibroin (SF) and phenylboronic acid/acrylamide for glucose-responsive insulin delivery. Six fabrication methods were investigated to maintain the glucose sensitivity of the hydrogel while avoiding deformation during fabrication. The optimized method was to fabricate smart MNs using a two-layer strategy, with a needle region formed by the SF combined semi-IPN hydrogel and the base layer fabricated by SF. The hybrid MN autonomously released insulin well-correspondent to the glucose change pattern via the regulation of the skin layer formed on the surface. Furthermore, this hybrid MN retained its original needle shape after 1 week in aqueous system, thus eliminating the safety concerns associated with dissolving MNs and suggesting the possibility for sustained delivery. This nondegradable smart MN is promising to provide on-demand insulin in a long-acting, painless, and convenient way. Field-effect transistor array modified by a stationary phase to generate
informative signal patterns for machine learning-assisted recognition of
gas-phase chemicals
Field-effect transistor array modified by a stationary phase to generate
informative signal patterns for machine learning-assisted recognition of
gas-phase chemicals
Yoshizumi, T.; Goda, T.; Yatabe, R.; Oki, A.; Matsumoto, A.; Oka, H.; Washio, T.; Toko, K.; Miyahara, Y.
Mol. Syst. Des. Eng. 2019, 4(2), 386-389.
AbstractWe propose an artificial intelligence-based chemical-sensing system integrating a porous gate field-effect transistor (PGFET) array modified by gas chromatography stationary phase materials and machine-learning techniques. The chemically sensitive PGFET array generates cross-reactive signals for computational analysis and shows potential for applications to compact intelligent sensing devices, including mobile electronic noses.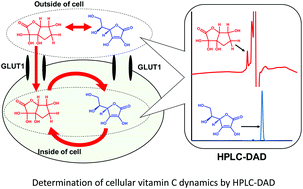 Determination of cellular vitamin C dynamics by HPLC-DAD
Determination of cellular vitamin C dynamics by HPLC-DAD
Miyazawa, T.; Matsumoto, A.; Miyahara, Y.
Analyst 2019, 144(11), 3483-3487.
Abstract/Cover A redox-sensitive inter-conversion between ascorbic acid (ASC) and its
oxidized form dehydroascorbic acid (DHA) in the intracellular
environment has been of exceptional interest to recent metabolomics and
pharmaceutical research. We developed a chromatographic protocol to
instantly determine these vitamers with each identity from cellular
extracts, without any labeling and pretreatments. Owing to its
simplicity, one can readily continue the assay for hours, an otherwise
difficult to cover timescale at which the intracellular DHA-ASC
conversion comes into play. The method was validated for the analysis of
pancreatic cancer cells, to our knowledge the first-ever study on a
nucleated cell type, to trace in detail their kinetics of glucose
transporter-dependent DHA uptake and, simultaneously, that for the
intracellular ASC conversion. The simplest of all the relevant
techniques and yet with the unique ability to provide each vitamer
identity on a high-throughput basis, this method should offer the most
practical option for VC-involved physiological and pharmaceutical
studies including high-dose VC cancer therapy.
A redox-sensitive inter-conversion between ascorbic acid (ASC) and its
oxidized form dehydroascorbic acid (DHA) in the intracellular
environment has been of exceptional interest to recent metabolomics and
pharmaceutical research. We developed a chromatographic protocol to
instantly determine these vitamers with each identity from cellular
extracts, without any labeling and pretreatments. Owing to its
simplicity, one can readily continue the assay for hours, an otherwise
difficult to cover timescale at which the intracellular DHA-ASC
conversion comes into play. The method was validated for the analysis of
pancreatic cancer cells, to our knowledge the first-ever study on a
nucleated cell type, to trace in detail their kinetics of glucose
transporter-dependent DHA uptake and, simultaneously, that for the
intracellular ASC conversion. The simplest of all the relevant
techniques and yet with the unique ability to provide each vitamer
identity on a high-throughput basis, this method should offer the most
practical option for VC-involved physiological and pharmaceutical
studies including high-dose VC cancer therapy.
Reviews
ボロノレクチンを利用したバイオエンジニアリング (Bioengineering using ’Borono-Lectin’)
松元 亮; 宮原 裕二.
医学のあゆみ (History of Medicine) 2019, 269(9), 697-702 (医歯薬出版株式会社).
Abstractボロン酸 (boronic acids)は糖との結合性から,レクチンになぞらえて“ボロノレクチン”と形容される.低分子ながら多様な生体分子と相互作用し,合成化学的にその選択性をデザインできるたぐいまれな分子群である.おもに有機合成におけるカップリング試薬としてのニーズ拡大に呼応し,今日ではきわめて多種多様な誘導体が安価に入手できる環境にある.ある局面ではリボースを至適に安定化することから,これを生命起源説のひとつである“RNA ワールド仮説”の支持根拠とする学説がある.ボロン酸が関わる分子認識では,それ自体の解離挙動と同期した顕著な親・疎水性変化が誘起され,これらを分子的にプログラムすることで高分子材料への複合的・階層的な環境応答性の付与,それを通じた機能化が可能となる.本稿では,著者らが展開する“ボロノレクチン”の機能をふんだんに生かした疾病診断や,ドラッグデリバリーシステム (DDS)関連の研究のなかから,とくにシアル酸認識によるがんターゲティングの手法,および糖尿病治療を目的としたグルコース応答型インスリン供給システムへの応用例を中心に概説する.「ボロノレクチン」工学が拓く新しい治療オプション (New treatment options developed through ’Borono-Lectin’ engineering)
松元 亮; 菅波 孝祥; 宮原 裕二.
MDB技術予測レポート ~2050年未来・世界を変える技術~ (MDB Technology Forecast Report - Technology that will change the future and the world in 2050) 2019, (株式会社日本能率協会総合研究所マーケティング・データ・バンク (MDB)).
 Determination of Intra- and Extra-cellular Vitamin C Dynamics
Determination of Intra- and Extra-cellular Vitamin C Dynamics
Miyazawa, T.; Matsumoto, A.; Miyahara, Y.
G.I.T. Laboratory Journal 2019.
AbstractThe redox-sensitive inter-conversion of ascorbic acid (ASC) and its oxidized form dehydroascorbic acid (DHA) in intracellular and extracellular environments is of exceptional interest at the forefront of metabolomics and pharmaceutical research, including high-dose vitamin C cancer therapy. A chromatographic protocol to instantly determine these vitamers in both forms from cellular extracts, without any labeling or pretreatment has been reported earlier.人工膵臓様デバイスによる血糖管理の未来 (The future of blood sugar management with artificial pancreas-like devices)
木村 真一郎; 田中 都; 松元 亮; 菅波 孝祥.
Pharma Medica 2019, 37(12), 59-62.
Biomaterials Science and Engineering in Japan: Attractive, Multidisciplinary, and Essential Research Field in Japan
Matsumoto, A.; Numata, K.
ACS Biomater. Sci. Eng. 2019, 5(11), 5559-5560.
’エレクトロニクスフリー’で完全合成型の人口膵臓の開発 (Development of electronics-free and completely synthetic artificial pancreas)
松元 亮.
バイオサイエンスとインダストリー (B&I) (Bioscience and Industry) 2019, 77(3), 256-257.
2018
 Enhanced Intracellular Delivery of siRNA by Controlling ATP-Responsivity
of Phenylboronic Acid-Functionalized Polyion Complex Micelles
Enhanced Intracellular Delivery of siRNA by Controlling ATP-Responsivity
of Phenylboronic Acid-Functionalized Polyion Complex Micelles
Naito, M.; Yoshinaga, N.; Ishii, T.; Matsumoto, A.; Miyahara, Y.; Miyata, K.; Kataoka, K.
Macromol. Biosci. 2018, 18(1), 1700357.
AbstractIntracellular delivery of small interfering RNA (siRNA) is a long-standing challenge in oligonucleotide therapeutics. Herein, adenosine triphosphate (ATP)-responsive polyion complex micelles assembled from poly(ethylene glycol)-block-poly(l-lysine) (PEG-PLys) bearing 4-carboxy-3-fluorophenylboronic acid (FPBA) moiety in the PLys side chains (FPBA micelle) for the delivery of cholesterol-modified siRNA (Chol-siRNA) are described. The pKa of FPBA moiety is 7.2 and, therefore, it exists in equilibrium between negatively charged tetravalent and noncharged hydrophobic trivalent forms in physiological pH conditions. Each form cooperatively stabilizes the micelle in distinct modes, that is, a covalent ester-linkage between charged boronate and ribose functionality at 3’ ends of Chol-siRNA and a hydrophobic interaction between noncharged boronic acid and Chol-siRNA. When exposed to ATP at a concentration associated with the intracellular environment, the Chol-siRNA/ boronate linkage is readily cleaved to facilitate the release of Chol-siRNA into cytoplasm. In order to further optimize this switching capability, the effect of FPBA modification rate is studied for the resulting ATP-responsive behavior of the micelles. As a result, the range of 23-35% in the modification rate is found suitable to maximize the gene silencing efficiency, demonstrating the potential of the FPBA-modified micelles as ATP-responsive smart siRNA carrier systems. Characterization and Optimization of Thermally Grown Iridium Oxide and
Its Application to pH Sensors
Characterization and Optimization of Thermally Grown Iridium Oxide and
Its Application to pH Sensors
Ratanaporncharoen, C.; Tabata, M.; Watanagool, N.; Goda, T.; Matsumoto, A.; Sriyudthsak, M.; Miyahara, Y.
Sens. Mater. 2018, 30(5), 1175-1185.
AbstractIn this study, we evaluate the thermal oxidization methods of iridium to simplify the fabrication of Ir/IrOx pH microsensors. Iridium oxide (IrOx) is a typical pH-sensing material. Specifically, three parameters of the previously reported “carbonate-melt” method are modified, namely, the oxidization temperature, oxidization time, and annealing process after oxidization. Then, we compare the characteristics of the fabricated sensors (i.e., the initial pH responsivity, time required to reach the steady state, and potential drift). The pH response depends on oxidization temperature and time.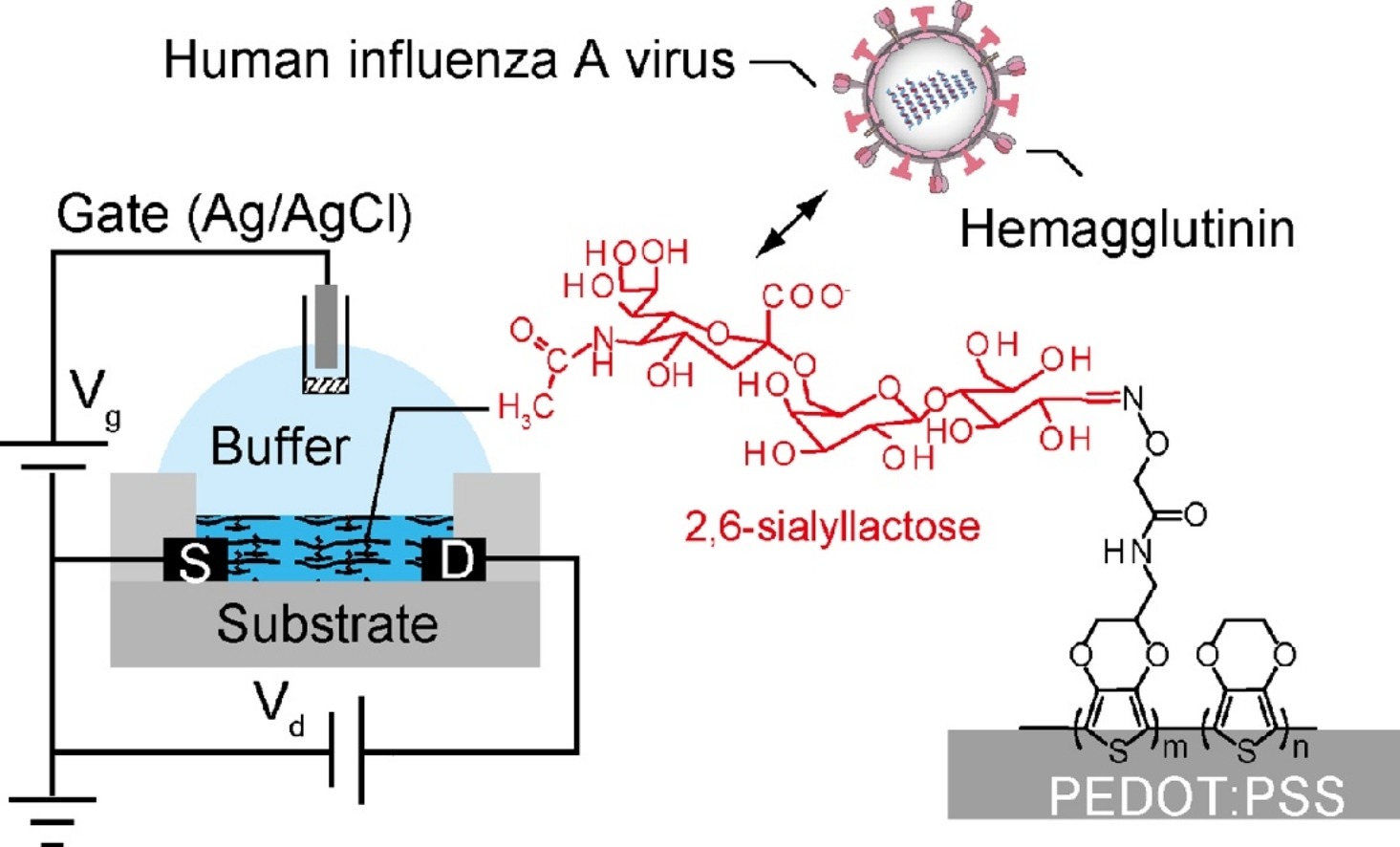 Human influenza virus detection using sialyllactose-functionalized
organic electrochemical transistors
Human influenza virus detection using sialyllactose-functionalized
organic electrochemical transistors
Hai, W.; Goda, T.; Takeuchi, H.; Yamaoka, S.; Horiguchi, Y.; Matsumoto, A.; Miyahara, Y.
Sens. Actuator B-Chem. 2018, 260, 635-641.
AbstractAn organic electrochemical transistor (OECT) with a trisaccharide-grafted conductive polymer channel was developed for human influenza A virus detection under aqueous conditions. A target recognition element was introduced into the electrochemical amplifier of the OECT for highly sensitive, selective, and label-free virus sensing. 3,4-Ethylenedioxythiophene (EDOT) and its derivative bearing an oxylamine moiety (EDOTOA) were electrochemically copolymerized on the channel region composed of a PEDOT:PSS thin film. The trisaccharides composed of Sia-alpha 2,6’-Gal-Glu (2,6-sialyllactose), a specific receptor for the hemagglutinin used as a spike protein on the surface of human influenza A virus, were covalently introduced into the EDOTOA unit. Changes in the drain current of the OECT were observed following virus adsorption onto the 2,6-sialyllactose-functionalized channel. A signal transduction mechanism involving a doping effect due to the adsorption of negatively-charged virus nanoparticles is proposed. The limit of detection was more than two orders of magnitude lower than commercial immunochromatographic influenza virus assays over the same detection time. Because of its processability with printing technologies and low power consumption, the OECT device developed here may be suitable for the wearable monitoring of influenza virus infection. (C) 2018 Elsevier B.V. All rights reserved.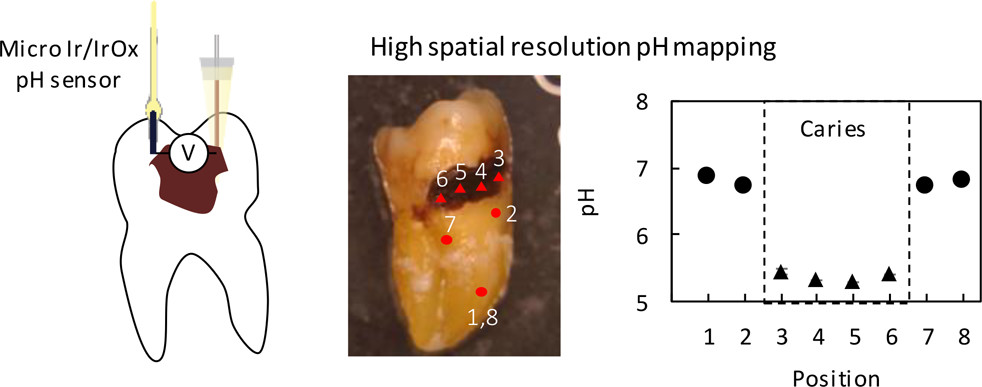 pH Mapping on Tooth Surfaces for Quantitative Caries Diagnosis Using
Micro Ir/IrOx pH Sensor
pH Mapping on Tooth Surfaces for Quantitative Caries Diagnosis Using
Micro Ir/IrOx pH Sensor
Ratanaporncharoen, C.; Tabata, M.; Kitasako, Y.; Ikeda, M.; Goda, T.; Matsumoto, A.; Tagami, J.; Miyahara, Y.
Anal. Chem. 2018, 90(7), 4925-4931.
AbstractA quantitative diagnostic method for dental caries would improve oral health, which directly affects the quality of life. Here we describe the preparation and application of Ir/IrOx pH sensors, which are used to measure the surface pH of dental caries. The pH level is used as an indicator to distinguish between active and arrested caries. After a dentist visually inspected and defined 18 extracted dentinal caries at various positions as active or arrested caries, the surface pH values of sound and caries areas were directly measured with an Ir/IrOx pH sensor with a diameter of 300 mu m as a dental explorer. The average pH values of the sound root, the arrested caries, and active caries were 6.85, 6.07, and 5.30, respectively. The pH obtained with an Ir/IrOx sensor was highly correlated with the inspection results by the dentist, indicating that the types of caries were successfully categorized. This caries testing technique using a micro Ir/IrOx pH sensor provides an accurate quantitative caries evaluation and has potential in clinical diagnosis.Gas-sensitive Field-effect Transistor Incorporating Polymer Layer and Porous Metal Electrode in the Gate Structure
Yoshizumi, T.; Goda, T.; Matsumoto, A.; Miyahara, Y.
Sens. Mater. 2018, 30(5, 1), 1001-1008.
AbstractField-effect transistor (FED-based sensors have attractive potential for use in fabricating miniaturized sensor arrays by semiconductor processes. In this work, a gas-sensitive FET incorporating a polyethylene glycol (PEG) film and porous Pt electrode in the gate structure is proposed. In this new type of gas-sensitive FET, the PEG layer provides a dielectric layer (i.e., a stationary phase) that interacts with gaseous molecules, while the porous Pt gate electrode allows gaseous molecules to effectively access the PEG layer. Features of the response pattern, such as peak height, recovery time, and peak shape, unique to the PEG-modified FET sensor were observed when exposed to vapor of volatile organic compounds (VOCs). The sensing mechanism of the gas-sensitive FET is discussed in terms of capacitance changes of the gate structure induced by gaseous molecule adsorption onto the stationary phase. A variety of polymeric materials might be used to modify the gate, and the proposed structure shows promise as a platform for cross-reactive FET-based gas sensor arrays for pattern recognition.
Reviews
困難な人工すい臓を実現した, 研究者の好奇心とは (What was the researcher’s curiosity that made the difficult artificial pancreas possible?)
松元 亮.
今日の健康 (Health Today) 2018, 2, 130-131.
Books
グルコース応答性ポリマーの設計と医療応用 (Design and medical applications of glucose-responsive polymers)
松元 亮; 菅波 孝祥; 宮原 裕二.
刺激応答性高分子ハンドブック ~分子応答性~ (Stimulus-responsive polymer handbook - Molecular responsiveness) 2018, Chapter 5.2, 678-684 (NTS Inc.).
2017
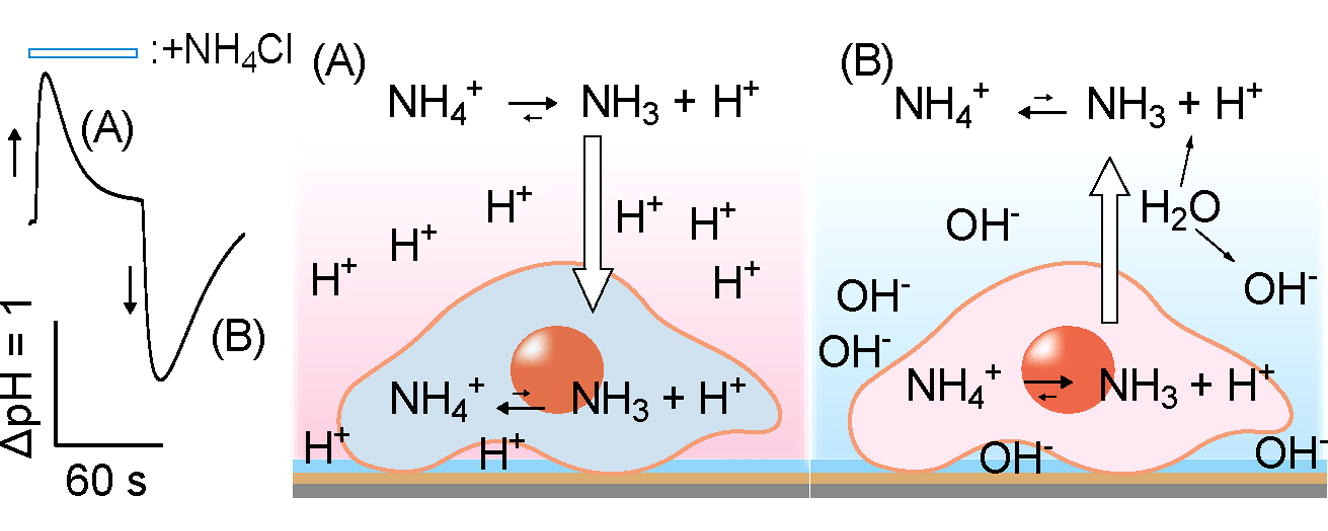 Proton-sensing transistor systems for detecting ion leakage from plasma
membranes under chemical stimuli
Proton-sensing transistor systems for detecting ion leakage from plasma
membranes under chemical stimuli
Imaizumi, Y.; Goda, T.; Schaffhauser, D. F.; Okada, J.; Matsumoto, A.; Miyahara, Y.
Acta Biomater. 2017, 50, 502-509.
AbstractThe membrane integrity of live cells is routinely evaluated for cytotoxicity induced by chemical or physical stimuli. Recent progress in bioengineering means that high-quality toxicity validation is required. Here, we report a pH-sensitive transistor system developed for the continuous monitoring of ion leakage from cell membranes upon challenge by toxic compounds. Temporal changes in pH were generated with high reproducibility via periodic flushing of HepG2 cells on a gate insulator of a proton-sensitive field-effect transistor with isotonic buffer solutions with without NH4Cl. The pH transients at the point of NH4Cl addition/withdrawal originated from the free permeation of NH3 across the semi-permeable plasma membranes, and the proton sponge effect produced by the ammonia equilibrium. Irreversible attenuation of the pH transient was observed when the cells were subjected to a membrane-toxic reagent. Experiments and simulations proved that the decrease in the pH transient was proportional to the area of the ion-permeable pores on the damaged plasma membranes. The pH signal was correlated with the degree of hemolysis produced by the model reagents. The pH assay was sensitive to the formation of molecularly sized pores that were otherwise not measurable via detection of the leakage of hemoglobin, because the hydrodynamic radius of hemoglobin was greater than 3.1 nm in the hemolysis assay. The pH transient was not disturbed by inherent ion-transporter activity. The ISFET assay was applied to a wide variety of cell types. The system presented here is fast, sensitive, practical and scalable, and will be useful for validating cytotoxins and nanomaterials. Direct and label-free influenza virus detection based on multisite
binding to sialic acid receptors
Direct and label-free influenza virus detection based on multisite
binding to sialic acid receptors
Horiguchi, Y.; Goda, T.; Matsumoto, A.; Takeuchi, H.; Yamaoka, S.; Miyahara, Y.
Biosens. Bioelectron. 2017, 92, 234-240.
AbstractA system to discriminate human or avian influenza A remains a highly sought-after tool for prevention of influenza pandemics in humans. Selective binding of the influenza A viral hemagglutinin (HA) to specific sialic acid (SA) receptors (Neu5Ac alpha(2-6)Gal in humans, Neu5Ac alpha(2-3)Gal in birds) is determined by the genotype of the HA and neuraminidase (NA) segments, making it one of the key characteristics that distinguishes human or avian influenza A virus. Here we demonstrate the direct detection of whole H1N1 influenza A virus using 6’-sialyllactose (Neu5Ac alpha(2-6)Gal beta(1-4)G1c, 6SL)-immobilized gold electrodes as biosensing surfaces. The sensitivity was higher than that of conventional immunochromatographic technique (ICT) for influenza virus and not restricted by genetic drift. The label-free detection technology via direct attachment of a whole virus using a chemically modified electrode is a promising means to provide a simple and rapid diagnostic system for viral infections.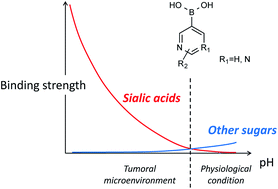 Heterocyclic boronic acids display sialic acid selective binding in a
hypoxic tumor relevant acidic environment
Heterocyclic boronic acids display sialic acid selective binding in a
hypoxic tumor relevant acidic environment
Matsumoto, A.; Stephenson-Brown, A. J.; Khan, T.; Miyazawa, T.; Cabral, H.; Kataoka, K.; Miyahara, Y.
Chem. Sci. 2017, 8(9), 6165-6170.
AbstractBoronic acids are well known for their ability to reversibly interact with the diol groups found in sugars and glycoproteins. However, they are generally indiscriminate in their binding. Herein we describe the discovery of a group of heterocyclic boronic acids demonstrating unusually high affinity and selectivity for sialic acids (SAs or N-acetylneuraminic acid), which are sugar residues that are intimately linked with tumor growth and cancer progression. Remarkably, these interactions strengthen under the weakly acidic pH conditions associated with a hypoxic tumoral microenvironment. In vitro competitive binding assays uncovered a significantly higher ability of 5-boronopicolinic acid, one of the derivatives identified in this work as a strong SA-binder, to interact with cell surface SA in comparison to a gold-standard structure, 3propionamidophenylboronic acid, which has proven to be an efficient SA-binder in numerous reports. This structure also proved to be suitable for further chemical conjugation with a well-preserved SA binding capability. These findings suggest an attractive alternative to other ongoing boronic acid based chemistry techniques aiming to achieve tumor-specific chemotherapies and diagnoses.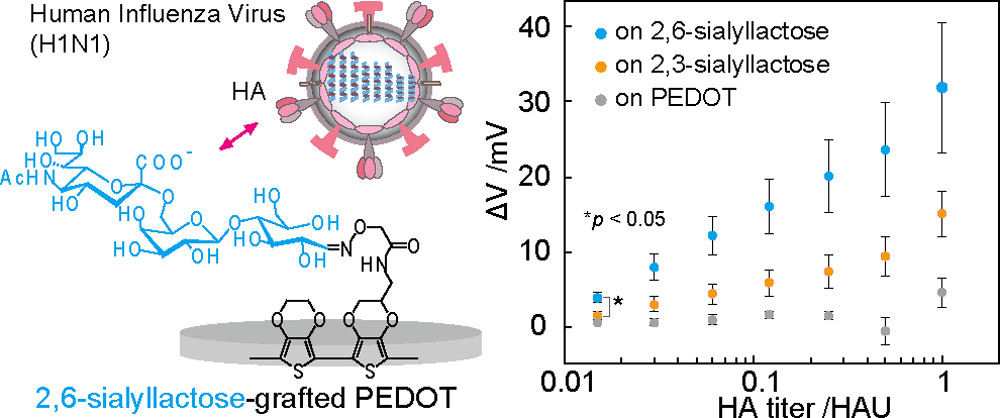 Specific Recognition of Human Influenza Virus with PEDOT Bearing Sialic
Acid-Terminated Trisaccharides
Specific Recognition of Human Influenza Virus with PEDOT Bearing Sialic
Acid-Terminated Trisaccharides
Hai, W.; Goda, T.; Takeuchi, H.; Yamaoka, S.; Horiguchi, Y.; Matsumoto, A.; Miyahara, Y.
ACS Appl. Mater. Interfaces 2017, 9(16), 14162-14170.
AbstractConducting polymers are good candidates for biosensor applications when molecular recognition element is imparted. We developed trisaccharide-grafted conducting polymers for label-free detection of the human influenza A virus (H1N1) with high sensitivity and specificity. A 3,4-ethylenedioxythiophene (EDOT) derivative bearing an oxylamine moiety was electrochemically copolymerized with EDOT. The obtained film was characterized by cyclic voltammetry, X-ray photoelectron spectroscopy, scanning electron microscopy, stylus surface profilometer, and AC-impedance spectroscopy. The trisaccharides comprising Sia-alpha 2,6’-Gal-Glu (2,6-sialyllactose) or Sia-alpha 2,3’-Gal-Glu (2,3sialyllactose) were covalently introduced to the side chain of the conducting polymers as a ligand for viral recognition. Immobilization of sialyllactose was confirmed by quartz crystal microbalance (QCM) and water contact angle measurements. Specific interaction of 2,6-sialyllactose with hemagglutinin in the envelope of the human influenza A virus (H1N1) was detected by QCM and potentiometry with enhanced sensitivity by 2 orders of magnitude when compared with that of commercially available kits. The developed conducting polymers possessing specific virus recognition are a good candidate material for wearable monitoring and point-of-care testing because of their processability and mass productivity in combination with printingDemonstration of thermo-sensitive tetra-gel with implication for facile and versatile platform for a new class of smart gels
Matsumoto, A.; Sato, S.; Sakamaki, T.; Sanjo, M.; Tabata, M.; Goda, T.; Asoh, T.-A.; Kikuchi, A.; Miyahara, Y.
J. Biomater. Sci.-Polym. Ed. 2017, 28(10-12), 1000-1009.
AbstractA tertiary branched poly(N-isopropylacrylamide) with controlled molecular weight, distribution and the end amino-functionalization (tetra-PNIPAAm-NH2) was studied for the ability to form a gel via in situ chain-end reaction with a counterpart tertiary branched poly(ethyleneglycol) bearing N-hydroxysuccinimide end groups (tetra-PEG-NHS), a well-documented class of building block to yield the tetra-gel. Some of these polymers, both comparable and distinct (relative to the counterpart) extended chain length pairs, provided a self-standing and macroscopically homogeneous gel, which was capable of undergoing thermo-sensitive and reversible change in hydration in line with the nature of PNIPAAm. Phantom network model based calculation indicated that a half molar fraction of the polymer chains in the network remained unreacted, revealing further room for optimizing the reaction condition. Since such tetra-PNIPAAm based motif can be readily tailored to a variety of other physicochemical stimuli-responsive analogues, our finding may give important insight into a platform for ‘smart’ tetra-gels with exceptional mechanical properties and potentially highly controllable molecular cut-off capability.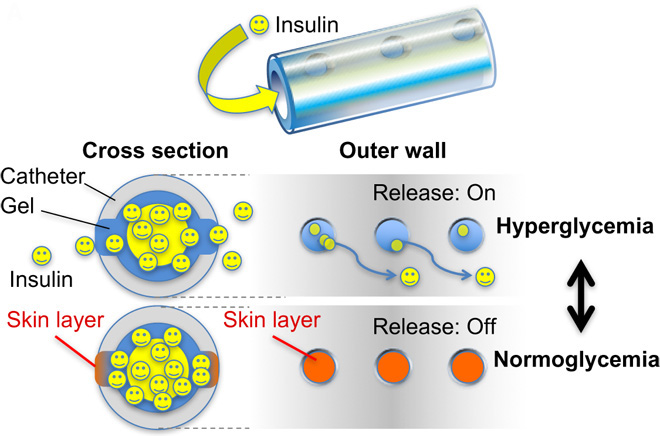 Synthetic “smart gel” provides glucose-responsive insulin delivery
in diabetic mice
Synthetic “smart gel” provides glucose-responsive insulin delivery
in diabetic mice
Matsumoto, A.; Tanaka, M.; Matsumoto, H.; Ochi, K.; Moro-oka, Y.; Kuwata, H.; Yamada, H.; Shirakawa, I.; Miyazawa, T.; Ishii, H.; Kataoka, K.; Ogawa, Y.; Miyahara, Y.; Suganami, T.
Sci. Adv. 2017, 3(11).
Abstract Press releaseAlthough previous studies have attempted to create electronics-free insulin delivery systemsusing glucose oxidase and sugar-binding lectins as a glucose-sensingmechanism, no successful clinical translation has hitherto been made. These protein-based materials are intolerant of long-term use and storage because of their denaturing and/or cytotoxic properties. We provide a solution by designing a protein-free and totally synthetic material-based approach. Capitalizing on the sugar-responsive properties of boronic acid, we have established a synthetic polymer gel-based insulin delivery device confined within a single catheter, which exhibits an artificial pancreas-like function in vivo. Subcutaneous implantation of the device in healthy and diabetic mice establishes a closed-loop system composed of “continuous glucose sensing” and “skin layer”-regulated insulin release. As a result, glucose metabolism was controlled in response to interstitial glucose fluctuation under both insulin-deficient and insulin-resistant conditions with at least 3-week durability. Our “smart gel” technology could offer a user-friendly and remarkably economic (disposable) alternative to the current state of the art, thereby facilitating availability of effective insulin treatment not only to diabetic patients in developing countries but also to those patients who otherwise may not be strongly motivated, such as the elderly, infants, and patients in need of nursing care.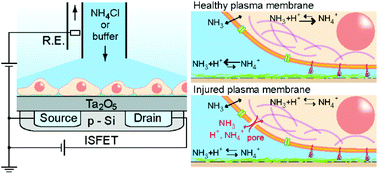 Identification of types of membrane injuries and cell death using whole
cell-based proton-sensitive field-effect transistor systems
Identification of types of membrane injuries and cell death using whole
cell-based proton-sensitive field-effect transistor systems
Imaizumi, Y.; Goda, T.; Matsumoto, A.; Miyahara, Y.
Analyst 2017, 142(18), 3451-3458.
AbstractAn NH4Cl-superfused system for a cell-cultured pH-sensing transistor was developed for detecting ion leakage across the plasma membranes of model HepG(2) cells. The screening of chemical species by the method developed and conventional membrane-leakage assays identified the types of membrane injuries: structural membrane disruption and pore formation. Apoptosis-mediated membrane disordering was detected by continuously monitoring the ion-barrier breakdown of the membranes using the transistor system for an extended period. Comparisons of the ISFET assay with conventional cytotoxicity assays distinguished the cell death by direct membrane injury from that by other organelle damage. Our cell-based transistor system is fast and sensitive to ion leakage of the plasma membrane due to the small hydrodynamic size of the proton and ammonium ions as the indicators. The combination of the ion leakage assay with the existing cytotoxicity assays is a new way of classifying membrane injury and cell death induced by external chemical stimuli.
Reviews
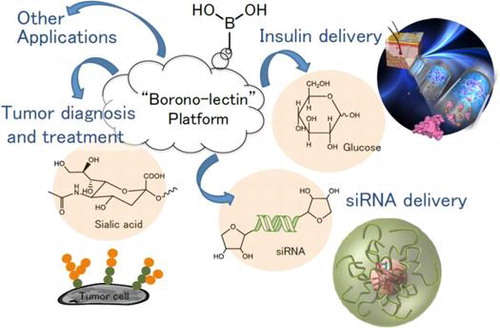 ‘Borono-lectin’ based engineering as a versatile platform for biomedical
applications
‘Borono-lectin’ based engineering as a versatile platform for biomedical
applications
Matsumoto, A.; Miyahara, Y.
Sci. Technol. Adv. Mater. 2017, 19(1), 18-30.
AbstractBoronic acids are well known for their ability to reversibly interact with the diol groups, a common motif of biomolecules including sugars and ribose. Due to their ability to interact with carbohydrates, they can be regarded as synthetic mimics of lectins, termed ‘borono-lectins’. The borono-lectins can be tailored to elicit a broad profile of binding strength and specificity. This special property has been translated into many creative biomedical applications in a way interactive with biology. This review provides a brief overview of recent efforts of polymeric materials-based engineering taking advantage of such virtue of ‘borono-lectins’ chemistry, related to the field of biomaterials and drug delivery applications.生体分子との相性が抜群な「ボロン酸工学」の発展を目指して (Aiming to develop ’boronic acid engineering’ that is highly compatible with biomolecules)
松元 亮.
Nanosky 2017, 3, 13.
2016
Potentiometric responses of ion-selective microelectrode with bovine serum albumin adsorption
Goda, T.; Yamada, E.; Katayama, Y.; Tabata, M.; Matsumoto, A.; Miyahara, Y.
Biosens. Bioelectron. 2016, 77, 208-214.
AbstractThere is a growing demand for an in situ measurement of local pH and ion concentrations in biological milieu to monitor ongoing process of bioreaction and bioresponse in real time. An ion-selective microelectrode can meet the requirements. However, the contact of the electrode with biological fluids induces biofouling by protein adsorption to result in a noise signal. Therefore, we investigated the relationship between the amount of nonspecific protein adsorption and the electrical signals in potentiometry by using ion-selective microelectrodes, namely silver/silver chloride (Ag/AgCl), iridium/iridium oxides (Ir/IrOx), and platinum/iridium oxides (Pt/IrOx). The microelectrodes reduced a potential change following the adsorption of bovine serum albumin (BSA) by comparison with the original metal microelectrodes without oxide layers. Suppression in the noise signal was attributed to the increased capacitance at the electrode/solution interface due to the formation of granulated metal oxide layer rather than a decrease in the amount of protein adsorbed. Ion sensitivity was maintained for Ir/IrOx against proton, but it was not for Ag/AgCI against chloride ion (Cl-), because of the interference of the equilibrium reaction by adsorbed BSA molecules on the electrode surface at <10(-2) M [Cl-] in the solution. The results open up the application of the Ir/IrOx microelectrode for measuring local pH in realistic dirty samples with a limited influence of electrode pollution by protein adsorption. (C) 2015 Elsevier B.V. All rights reserved.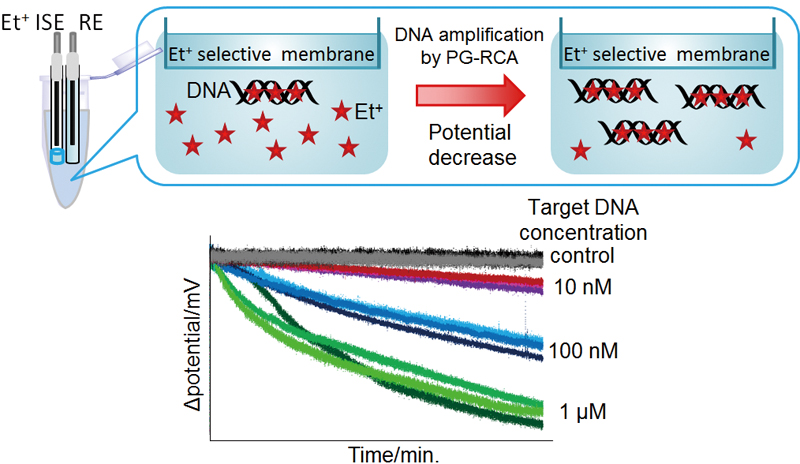 Real-time Monitoring and Detection of Primer Generation-Rolling Circle
Amplification of DNA Using an Ethidium Ion-selective Electrode
Real-time Monitoring and Detection of Primer Generation-Rolling Circle
Amplification of DNA Using an Ethidium Ion-selective Electrode
Seichi, A.; Kozuka, N.; Kashima, Y.; Tabata, M.; Goda, T.; Matsumoto, A.; Iwasawa, N.; Citterio, D.; Miyahara, Y.; Suzuki, K.
Anal. Sci. 2016, 32(5), 505-510.
AbstractAn electrochemical detection system for an isothermal DNA amplification method using an ion-selective electrode (ISE) was developed as a low-cost, simple and real-time monitoring system. The system is based on potentiometry using an ethidium ion (Et+) selective electrode that relies on monitoring DNA amplification by measuring potential changes in the reaction solution containing ethidium bromide (EtBr) as an intercalator to DNA. With progressing primer generation rolling circle amplification (PG-RCA) under isothermal condition at 37 degrees C, EtBr is bound to the newly formed DNA, resulting in a lowered free EtBr concentration in the sample solution. In this case, the Et+ ISE potential allows real-time monitoring of the PG-RCA reaction in the range of 10 nM- 1 mu M initial target DNA. Boronate-functionalized Polymer Gel-based Insulin Delivery System with
Improved Stability in Performance: A Comparative Structure-Function
Study
Boronate-functionalized Polymer Gel-based Insulin Delivery System with
Improved Stability in Performance: A Comparative Structure-Function
Study
Matsumoto, A.; Yuasa, M.; Matsumoto, H.; Sanjo, M.; Tabata, M.; Goda, T.; Hoshi, T.; Aoyagi, T.; Miyahara, Y.
Chem. Lett. 2016, 45(4), 460-462.
AbstractIn an attempt to develop a totally synthetic, self-regulated insulin delivery system to treat diabetes, N-substituted poly(acrylamide) copolymer gels bearing boronate functionality have been chemically optimized so as to undergo an abrupt and glucose dependent change in hydration. This study highlights an importance of fine-tuning the mesh size of the gel relative to that of insulin in order to achieve the better controlled-release in a manner less sensitive to the temperature, even with an apparently marginal change of hydration.Miniaturized Ir/IrOx pH Sensor for Quantitative Diagnosis of Dental Caries
Tabata, M.; Ratanaporncharoen, C.; Asano, A.; Kitasako, Y.; Ikeda, M.; Goda, T.; Matsumoto, A.; Tagami, J.; Miyahara, Y.
Procedia Eng. 2016, 168, 598-601.
AbstractDiagnosis of dental caries is one of the most basic skills for oral healthcare professionals; however, the detection methods are based on visually diagnosis, such as inspection, palpation, and X-ray imaging. Therefore, quantitative and objective diagnostic technique is needed [1]. The aim of this study is fabrication and evaluation of Iridium/Iridium Oxide (Ir/IrOx) pH sensor for quantitative diagnosis of dental caries. The potentiometric response to pH showed -57.4mV/pH in evaluated pH range from 4 to 8, which demonstrated that our electrode possesses the excellent proton sensitivity (Nernst slope; -59.2mV/pH). Based on the evaluation results of dental caries, we confirmed the possibility for quantitative diagnosis of dental caries using Ir/IrOx electrode based on the pH as an index.Label-free and Electrochemical Detection of Nucleic Acids Based on Isothermal Amplification in Combination with Solid-state pH Sensor
Tabata, M.; Katayama, Y.; Mannan, F.; Seichi, A.; Suzuki, K.; Goda, T.; Matsumoto, A.; Miyahara, Y.
Procedia Eng. 2016, 168, 419-422.
AbstractLabel-free quantification methods for nucleic acids are attracting attention, since they provide potential tools in clinical applications such as disease diagnosis or prognosis observation. In this research, we developed a simpler and cost-effective electrical monitoring device of nucleic acid amplification, combining an isothermal amplification method and an Iridium/Iridium oxide (Ir/IrOx) electrode without labeling. The fabricated Ir/IrOx electrode showed ideal Nernstian response under the various pH buffer solutions, and its potential response was less affected even in the presence of charged proteins. Moreover, we successfully real-time monitored primer generation-rolling circle amplification (PG-RCA) at the 0, 10, 100, 1000 pM of target DNA by detection of released proton during amplification reaction at the constant reaction temperature. This label-free and portable device might be received considerable attention as a useful platform for a point-of-care testing in clinical use.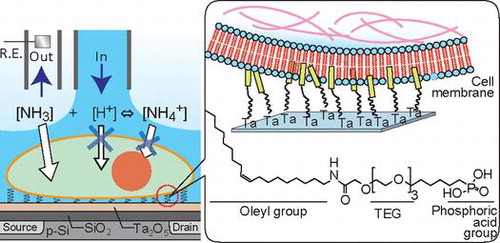 Oleyl group-functionalized insulating gate transistors for measuring
extracellular pH of floating cells
Oleyl group-functionalized insulating gate transistors for measuring
extracellular pH of floating cells
Imaizumi, Y.; Goda, T.; Toya, Y.; Matsumoto, A.; Miyahara, Y.
Sci. Technol. Adv. Mater. 2016, 17(1), 337-345.
AbstractThe extracellular ionic microenvironment has a close relationship to biological activities such as by cellular respiration, cancer development, and immune response. A system composed of ion-sensitive field-effect transistors (ISFET), cells, and program-controlled fluidics has enabled the acquisition of real-time information about the integrity of the cell membrane via pH measurement. Here we aimed to extend this system toward floating cells such as T lymphocytes for investigating complement activation and pharmacokinetics through alternations in the plasma membrane integrity. We functionalized the surface of tantalum oxide gate insulator of ISFET with oleyl-tethered phosphonic acid for interacting with the plasma membranes of floating cells without affecting the cell signaling. The surface modification was characterized by X-ray photoelectron spectroscopy and water contact angle measurements. The Nernst response of -37.8 mV/pH was obtained for the surface-modified ISFET at 37 degrees C. The oleyl group-functionalized gate insulator successfully captured Jurkat T cells in a fluidic condition without acute cytotoxicity. The system was able to record the time course of pH changes at the cells/ISFET interface during the process of instant addition and withdrawal of ammonium chloride. Further, the plasma membrane injury of floating cells after exposure by detergent Triton (TM) X-100 was successfully determined using the modified ISFET with enhanced sensitivity as compared with conventional hemolysis assays. [GRAPHICS]
Books
Field-Effect Transistors for Detection of Biomolecular Recognition
Tabata, M.; Goda, T.; Matsumoto, A.; Miyahara, Y.
Intelligent Nanosystems for Energy, Information and Biological Technologies 2016, 13-25 (Springer).
AbstractElectrical biosensors have attracted increasing attention in such fields as point-of-care testing, drug discovery, and healthcare products. In order for next-generation biosensor platforms to become more useful in our daily lives, it will be necessary to significantly improve their sensitivity, specificity, and parallelism. A precisely designed thin layer in molecular dimension on a solid substrate is essential for biosensing. The surfaces of biosensors are designed to capture target bioanalytes. In addition, the solid/liquid interface plays an important role in realizing additional functionalities such as target manipulation, signal stabilization, and switching. A functional interface combined with a field-effect device would enable on-demand label-free biosensing in a portable format. In this chapter, we provide an overview of biomolecular recognition in the context of electrochemical sensing and biosensing. Also, we review recent progress and trends in biosensing, including our own research.
2015
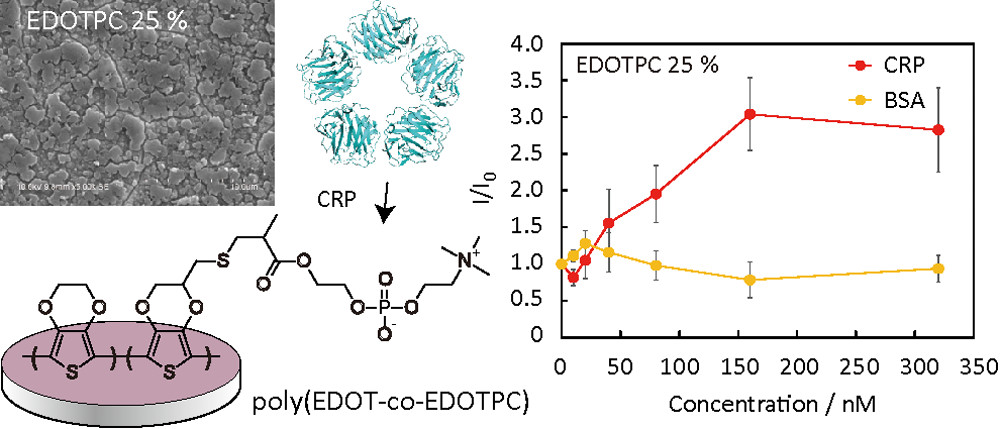 Poly(3,4-ethylenedioxythiophene) Bearing Phosphorylcholine Groups for
Metal-Free, Antibody-Free, and Low-Impedance Biosensors Specific for
C-Reactive Protein
Poly(3,4-ethylenedioxythiophene) Bearing Phosphorylcholine Groups for
Metal-Free, Antibody-Free, and Low-Impedance Biosensors Specific for
C-Reactive Protein
Goda, T.; Toya, M.; Matsumoto, A.; Miyahara, Y.
ACS Appl. Mater. Interfaces 2015, 7(49), 27440-27448.
AbstractConducting polymers possessing biorecognition elements are essential for developing electrical biosensors sensitive and specific to clinically relevant biomolecules. We developed a new 3,4-ethylenedioxythiophene (EDOT) derivative bearing a zwitterionic phosphorylcholine group via a facile synthesis through the Michael-type addition thiol-ene “click” reaction for the detection of an acute-phase biomarker human C-reactive protein (CRP). The phosphorylcholine group, a major headgroup in phospholipid, which is the main constituent of plasma membrane, was also expected to resist nonspecific adsorption of other proteins at the electrode/solution interface. The biomimetic EDOT derivative was randomly copolymerized with EDOT, via an electropolyrnerization technique with a dopant sodium perchlorate, onto a glassy carbon electrode to make the synthesized polymer film both conductive and target-responsive. The conducting copolymer films were characterized by cyclic voltammetry, scanning electron microscopy, attenuated total reflection Fourier transform infrared spectroscopy, X-ray photoelectron spectroscopy, and electrochemical impedance spectroscopy. The specific interaction of CRP with phosphorylcholine in a calcium-containing buffer solution was determined by differential pulse voltammetry, which measures the altered redox reaction between the indicators ferricyanide/ferrocyanide as a result of the binding event. The conducting polymer-based protein sensor achieved a limit of detection of 37 nM with a dynamic range of 10-160 nM, covering the dynamically changing CRP levels in circulation during the acute phase. The results will enable the development of metal-free, antibody-free, and low-impedance electrochemical biosensors for the screening of nonspecific biomarkers of inflammation and infection.Dual aptamer-immobilized surfaces for improved affinity through multiple target binding in potentiometric thrombin biosensing
Goda, T.; Higashi, D.; Matsumoto, A.; Hoshi, T.; Sawaguchi, T.; Miyahara, Y.
Biosens. Bioelectron. 2015, 73, 174-180.
AbstractWe developed a label-free and reagent-less potentiometric biosensor with improved affinity for thrombin. Two different oligomeric DNA aptamers that can recognize different epitopes in thrombin were introduced in parallel or serial manners on the sensing surface to capture the target via multiple contacts as found in many biological systems. The spacer and linker in the aptamer probes were optimized for exerting the best performance in molecular recognition. To gain the specificity of the sensor to the target, an antifouling molecule, sulfobeaine-3-undecanethiol (SB), was introduced on the sensor to form a self-assembled monolayer (SAM). Surface characterization revealed that the aptamer probe density was comparable to the distance of the two epitopes in thrombin, while the backfilling SB SAM was tightly aligned on the surface to resist nonspecific adsorption. The apparent binding parameters were obtained by thrombin sensing in potentiometry using the 1:1 Langmuir adsorption model, showing the improved dissociation constants (K-d) with the limit of detection of 5.5 nM on the dual aptamer-immobilized surfaces compared with single aptamer-immobilized ones. A fine control of spacer and linker length in the aptamer ligand was essential to realize the multivalent binding of thrombin on the sensor surface. The findings reported herein are effective for improving the sensitivity of potentiometric biosensor in an affordable way towards detection of tiny amount of biomolecules. (C) 2015 Elsevier B.V. All rights reserved.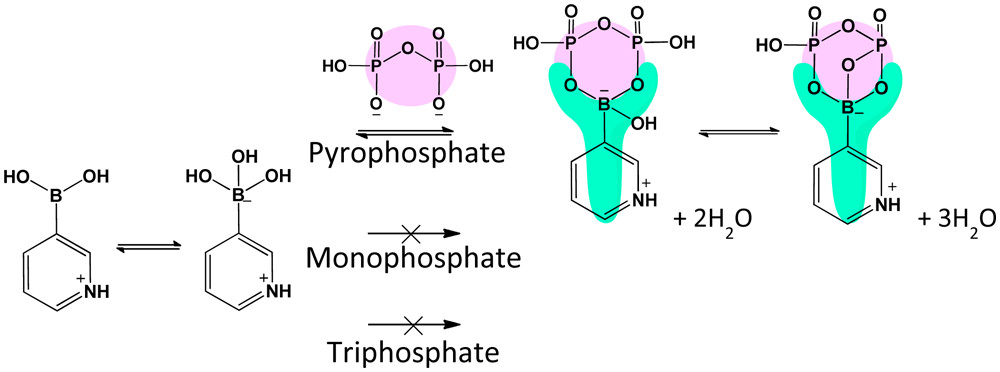 Boronate Based Metal-Free Platform for Diphosphate-Specific Molecular
Recognitions
Boronate Based Metal-Free Platform for Diphosphate-Specific Molecular
Recognitions
Sanjoh, M.; Iizuka, D.; Matsumoto, A.; Miyahara, Y.
Org. Lett. 2015, 17(3), 588-591.
AbstractA reversible boronate-diol interaction provides a versatile synthetic platform for molecular recognitions whose binding specificity can be molecularly tailored. We found that boronate derivatives with relatively strong acidity generally undergo a diphosphate-specific recognition among other phosphates under weakly acidic pH conditions, a feature relevant to DNA sequencing. B-11 and P-31 NMR studies identified “tetrahedral boronate and divalent diphosphate” as a pair responsible for forming a 1:1 stoichiometric complex, which manifests as a unique pH-dependent stability.
2014
Chemical Optimization for Simultaneous Voltammetric Detection of Molybdenum and Silver Nanoparticles in Aqueous Buffer Solutions
Goda, T.; Oohashi, M.; Matsumoto, A.; Hoshi, T.; Sawaguchi, T.; Pumera, M.; Miyahara, Y.
ChemElectroChem 2014, 1(12), 2110-2115.
AbstractSimultaneous acquisition of redox signals from different types of metal nanoparticles (NPs) in a multiplexing system requires a good separation in redox potential from each component for successful identification. The appearance of a single distinct peak for each NP type is also preferred. Here, we report variations in the electrochemical nature of molybdenum (Mo) and silver (Ag) NPs cast on a glassy carbon electrode that are sensitive to various conditions of the measurement solution, such as buffering capacity and electrolyte concentration. A series of experiments allowed us to find optimal conditions, where Mo NPs showed a single oxidation peak with good separation of the redox potential from that of Ag NPs (Delta E approximate to 0.25 V), by using differential-pulse voltammetry (DPV). In the simultaneous monitoring of Mo and Ag NPs under the optimal conditions, the peak currents in DPV were quantitative for the amount of Mo/Ag NPs ranging from 7.8/20 to 500/20 ngng(-1) and from 500/20 to 500/0.31 ngng(-1).Fabrication of Self-assembled Monolayer/AgCl Mixed Surface and It’s Electrochemical Properties
Tabata, M.; Nogami, K.; Goda, T.; Matsumoto, A.; Miyahara, Y.
IEEJ Trans. Sens. Micromachines 2014, 134(10), 315-319.
AbstractThere is increasing interest on solid-state biosensors due to potential advantages such as small size, low-cost, high density integration for point of care testing and next-generation drug discovery. In many cases, a gold electrode is used as a sensing surface on which specific biomolecule recognition events take place. However, gold is not always the best material for potentiometric measurement since the surface of gold electrode is polarized in aqueous solution, and there is room for improvement in stability of interface potential at the gold electrode surface. In this paper, we fabricated and characterized the mixed surface of self-assembled monolayer of alkanethiol and silver chloride to realize both stable interface potential and immobilization of biomolecules.Chemo-Electrical Signal Transduction by Using Stimuli-Responsive Polymer Gate-Modified Field Effect Transistor
Matsumoto, A.; Tsurui, Y.; Matsumoto, H.; Maeda, Y.; Hoshi, T.; Sawaguchi, T.; Miyahara, Y.
Chemosensors 2014, 2(2), 97-107.
AbstractA glucose-responsive polymer brush was designed on a gold electrode and exploited as an extended gate for a field effect transistor (FET) based biosensor. A permittivity change at the gate interface due to the change in hydration upon specific binding with glucose was detectable. The rate of response was markedly enhanced compared to the previously studied cross-linked or gel-coupled electrode, owing to its kinetics involving no process of the polymer network diffusion. This finding may offer a new strategy of the FET-based biosensors effective not only for large molecules but also for electrically neutral molecules such as glucose with improved kinetics.Current Development Status and Perspectives of Self-Regulated Insulin Delivery Systems: A Review
Matsumoto, A.; Miyahara, Y.
Electr. Commun. Jpn. 2014, 97(12), 57-61.
AbstractDiabetes is not an infectious disease but its increasingly rapid and worldwide spread has been recognized as pandemic. Despite the necessity for continuous and accurate glycemic control in the management of insulin-dependent diabetes mellitus, the current palliative treatment relies almost solely on patient self-injection of insulin, which not only impinges on the QOL of patients but also fails to precisely control the dose of insulin, where an overdose must be strictly avoided since it can otherwise cause serious hypoglycemia. The development of self-regulated insulin delivery systems is a long-standing challenge for materials science, for which the exploitation of glucose oxidase and sugar-binding lectin are two prevalent strategies to achieve the function of glucose sensitivity. These protein-based components, however, are inappropriate for long-term use and storage due to their denaturing and cytotoxic natures, and they are hardly suitable for any implantable applications. Thus, they have not yet been put in clinical use to date. This review intends to provide an overview of the current efforts to develop a thoroughly synthetic alternative making use of a phenylboronic acid-derived polyacrylamide gel.
Reviews
Phenylboronic Acids-based Diagnostic and Therapeutic Applications
Sanjoh, M.; Miyahara, Y.; Kataoka, K.; Matsumoto, A.
Anal. Sci. 2014, 30(1), 111-117.
AbstractPhenylboronic acid (PBA) derivatives are known to form reversible complexes with polyols, including sugars. This unique chemistry has provided many useful molecular bases for analytical and therapeutic applications. This mini-review highlights some new aspects of related research efforts with a special focus on the interaction with sialic acid as a new class of molecular targets and other PBA-based strategies for drug delivery applications.New directions in the design of phenylboronate-functionalized polymers for diagnostic and therapeutic applications
Matsumoto, A.; Kataoka, K.; Miyahara, Y.
Polym. J. 2014, 46(8), 483-491.
AbstractPhenylboronic acid (PBA) derivatives, which are often termed boronolectins,’ are regarded as a synthetic mimic of lectins because of their ability to interact with various carbohydrates. This unique chemistry has already borne fruit as the molecular bases for glucose sensors and some bio-separation applications. This focus review highlights some of the emerging PBA-based research directions toward more versatile diagnostic and therapeutic targets, which the authors are currently pursuing. The topics reviewed include PBA-mediated targeting to sialic acid (SA) as a methodology relevant to tumor diagnosis and treatment. Other drug delivery systems, including those for siRNA and insulin, in which PBA has a unique role in physicochemical signal transduction, will also be summarized.
Books
スマートゲルを利用した完全合成型の“人工膵臓 (Development of a Totally Synthetic ’Artificial Pancreas’)
松元 亮; 宮原 裕二.
スマート・ヒューマンセンシング ~健康ビッグデータ時代のためのセンサ・情報・エネルギー技術~ (Human Sensing for Smart Life Care - Sensor, Information & Energy Technologies on Health Conscious Society with Big Data) 2014, Chapter 3.4, 233-240 (CMC Publishing Co., Ltd).
ボロン酸ゲルによるインスリンデリバリーシステム (Phenylboronate Gel Based Insulin Delivery Systems)
松元 亮; 片岡一則; 宮原 裕二.
ゲルテクノロジーハンドブックグ ~機能設計・評価・シミュレーションから製造プロセス・製品化まで~ (Science and Technology Handbook of Gels - From functional design, evaluation, and simulation to manufacturing process and commercialization) 2014, Chapter 3.1, (NTS Inc.).
機能性超分子界面を用いるバイオトランジスタ (Biotransistors with Supermolecular Interface)
宮原 裕二; 松元 亮; 合田 達郎; 前田 康弘; 田畑 美幸; 三條 舞.
超分子材料の設計と応用展開 2014, Chapter 2.1.3, 13-25 (CMC Publishing Co., Ltd).
2013
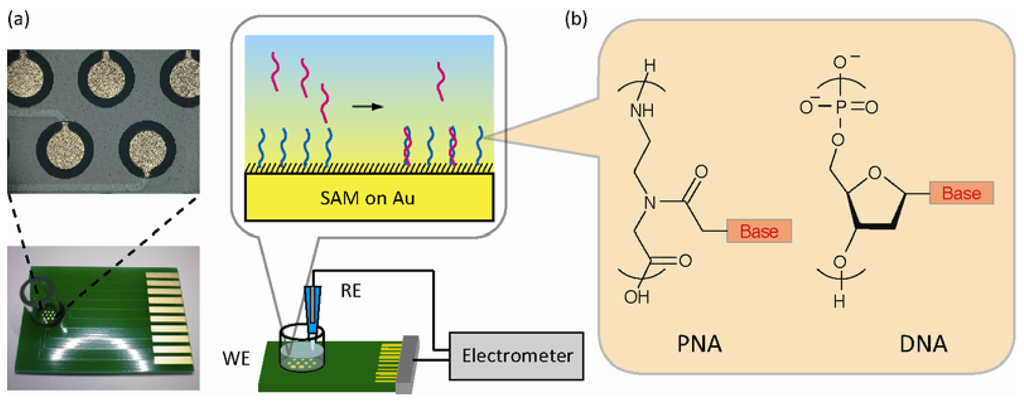 Label-Free Potentiometry for Detecting DNA Hybridization Using Peptide
Nucleic Acid and DNA Probes
Label-Free Potentiometry for Detecting DNA Hybridization Using Peptide
Nucleic Acid and DNA Probes
Goda, T.; Singi, A. B.; Maeda, Y.; Matsumoto, A.; Torimura, M.; Aoki, H.; Miyahara, Y.
Sensors 2013, 13(2), 2267-2278.
AbstractPeptide nucleic acid (PNA) has outstanding affinity over DNA for complementary nucleic acid sequences by forming a PNA-DNA heterodimer upon hybridization via Watson-Crick base-pairing. To verify whether PNA probes on an electrode surface enhance sensitivity for potentiometric DNA detection or not, we conducted a comparative study on the hybridization of PNA and DNA probes on the surface of a 10-channel gold electrodes microarray. Changes in the charge density as a result of hybridization at the solution/electrode interface on the self-assembled monolayer (SAM)-formed microelectrodes were directly transformed into potentiometric signals using a high input impedance electrometer. The charge readout allows label-free, reagent-less, and multi-parallel detection of target oligonucleotides without any optical assistance. The differences in the probe lengths between 15- to 22-mer dramatically influenced on the sensitivity of the PNA and DNA sensors. Molecular type of the capturing probe did not affect the degree of potential shift. Theoretical model for charged rod-like duplex using the Gouy-Chapman equation indicates the dominant effect of electrostatic attractive forces between anionic DNA and underlying electrode at the electrolyte/electrode interface in the potentiometry.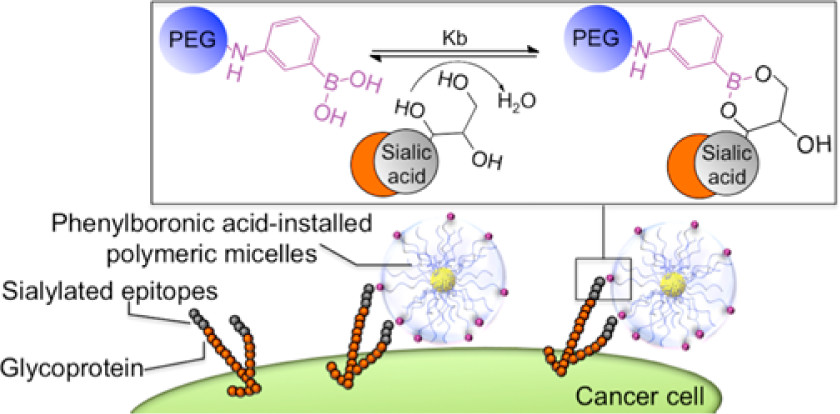 Phenylboronic Acid-Installed Polymeric Micelles for Targeting Sialylated
Epitopes in Solid Tumors
Phenylboronic Acid-Installed Polymeric Micelles for Targeting Sialylated
Epitopes in Solid Tumors
Deshayes, S.; Cabral, H.; Ishii, T.; Miura, Y.; Kobayashi, S.; Yamashita, T.; Matsumoto, A.; Miyahara, Y.; Nishiyama, N.; Kataoka, K.
J. Am. Chem. Soc. 2013, 135(41), 15501-15507.
AbstractLigand-mediated targeting of nanocarriers to tumors is an attractive strategy for increasing the efficiency of chemotherapies. Sialylated glycans represent a propitious target as they are broadly overexpressed in tumor cells. Because phenylboronic acid (PBA) can selectively recognize sialic acid (SA), herein, we developed PBA-installed micellar nanocarriers incorporating the parent complex of the anticancer drug oxaliplatin, for targeting sialylated epitopes overexpressed on cancer cells. Following PBA-installation, the micelles showed high affinity for SA, as confirmed by fluorescence spectroscopy even at intratumoral pH conditions, i.e., pH 6.5, improving their cellular recognition and uptake and enhancing their in vitro cytotoxicity against B16F10 murine melanoma cells. In vivo, PBA-installed micelles effectively reduced the growth rate of both orthotopic and lung metastasis models of melanoma, suggesting the potential of PBA-installed nanocarriers for enhanced tumor targetingBiotransistor with functional nanointerface containing biomolecules
Tabata, M.; Goda, T.; Matsumoto, A.; Miyahara, Y.
IEEJ Trans. Sens. Micromachines 2013, 133(11), B314-B319.
AbstractThe convergence of nano-/bio- technology with biomedicine has occurred tremendously over the last few decades. In particular, next-generation biosensors offer great potential to treat serious medical problems in the fields of diagnostics, therapy, and drug delivery. Advantageously, an electrical biosensor platform composed of target-capturing element and semiconductor device as a signal transducer is compatible with a high-throughput analysis via miniaturization and integration into a small chip for decentralized bioanalytical applications. So far, we have successfully achieved potentiometric biosensing based mostly on a direct readout of innately charged biomolecules using field effect transistor (FET). Highly specific molecular recognition is available by constructing engineered biointerface on the sensor. Furthermore, the potentiometric technique provides the ability to perform label-free and real-time detection for various biomolecules. These features would help to promote personalized medicine for individual patients in the future. Here we describe fundamental aspects of electrical biosensing and review recent developments of the FET-based biotransistors.Simple and robust strategy for potentiometric detection of glucose using fluorinated phenylboronic acid self-assembled monolayer
Matsumoto, A.; Matsumoto, H.; Maeda, Y.; Miyahara, Y.
Biochim. Biophys. Acta-Gen. Subj. 2013, 1830(9, SI), 4359-4364.
AbstractBackground: Field effect transistor (PET) based signal-transduction (Bio-FET) is an emerging technique for label-free and real-time basis biosensors for a wide range of targets. Glucose has constantly been of interest due to its clinical relevance. Use of glucose oxidase (GOD) and a lectin protein Concanavalin A are two common strategies to generate glucose-dependent electrochemical events. However, these protein-based materials are intolerant of long-term usage and storage due to their inevitable denaturing. Methods: A phenylboronic acid (PEA) modified self-assembled monolayer (SAM) on a gold electrode with an optimized disassociation constant of PBA, that is, 3-fluoro-4-carbamoyl-PBA possessing its pKa of 7.1, was prepared and utilized as an extended gate electrode for Bio-FET. Results: The prepared electrode showed a glucose-dependent change in the surface potential under physiological conditions, thus providing a remarkably simple rationale for the glyco-sensitive Bio-FET. Importantly, the PBA modified electrode showed tolerance to relatively severe heat and drying treatments; conditions under which protein based materials would surely be denatured. Conclusions: A PEA modified SAM with optimized disassociation constant (pKa) can exhibit a glucose-dependent change in the surface potential under physiological conditions, providing a remarkably simple but robust method for the glyco-sensing. General significance: This protein-free, totally synthetic glyco-sensing strategy may offer cheap, robust and easily accessible platform that may be useful in developing countries. This article is part of a Special Issue entitled Organic Bioelectronics-Novel Applications in Biomedicine. (C) 2013 Elsevier B.V. All rights reserved.
Reviews
生体計測技術 ~バイオセンシング~ (Biological Measurement Technology - Biosensing -)
松元 亮; 宮原 裕二.
人工臓器 (Artificial Organs) 2013, 42(3), 213-216.
自律型インスリン投与デバイスを目指したグルコース応答ゲルの開発 (Glucose-Responsive Gel for Self-Regulated Insulin Delivery System)
松元 亮; 石井 武彦; 片岡 一則; 宮原 裕二.
Drug Deliv. Syst. 2013, 28(2), 119-126.
Abstract今日, インスリン依存型糖尿病の対症療法としては, 患者自身によるインスリン頻回注射がもっとも一般的である。しかしながら, この方法は, 患者の生活の質 (QOL: Quality of Life)を著しく損なううえ, 正確な用量設定も困難であり, 長期的な血糖値管理の目的を満足するものではない。このような背景から, グルコースオキシダーゼやレクチンを利用して自律型 (フィードバック型)のインスリン供給システム, いわば“人工膵臓”を工学的につくる試みが多くなされてきた。ところが, これらタンパク質由来の物質は, 変性や細胞毒性のために長期使用や保管が困難であり, いずれも実用には至っていない。本稿では, 合成分子を組み上げて得られる「ボロン酸ゲル」を用いてこれを代替する筆者らの取り組みを中心に概説する。 Current and emerging challenges of field effect transistor based
bio-sensing
Current and emerging challenges of field effect transistor based
bio-sensing
Matsumoto, A.; Miyahara, Y.
Nanoscale 2013, 5(22), 10702-10718.
AbstractField-effect-transistor (FET) based electrical signal transduction is an increasingly prevalent strategy for bio-sensing. This technique, often termed ’Bio-FETs’, provides an essentially label-free and real-time based bio-sensing platform effective for a variety of targets. This review highlights recent progress and challenges in the field. A special focus is on the comprehension of emerging nanotechnology-based approaches to facilitate signal-transduction and amplification. Some new targets of Bio-FETs and the future perspectives are also discussed.機能性ナノ界面を有するバイオトランジスタ (Biotransistor with Functional Nanointerface Containing Biomolecules)
田畑 美幸; 合田 達郎; 松元 亮; 宮原 裕二.
電気学会論文誌E (センサ・マイクロマシン部門誌) (IEEJ Transactions on Sensors and Micromachines) 2013, 133(11), 314-319.
AbstractThe convergence of nano-/bio- technology with biomedicine has occurred tremendously over the last few decades. In particular, next-generation biosensors offer great potential to treat serious medical problems in the fields of diagnostics, therapy, and drug delivery. Advantageously, an electrical biosensor platform composed of target-capturing element and semiconductor device as a signal transducer is compatible with a high-throughput analysis via miniaturization and integration into a small chip for decentralized bioanalytical applications. So far, we have successfully achieved potentiometric biosensing based mostly on a direct readout of innately charged biomolecules using field effect transistor (FET). Highly specific molecular recognition is available by constructing engineered biointerface on the sensor. Furthermore, the potentiometric technique provides the ability to perform label-free and real-time detection for various biomolecules. These features would help to promote personalized medicine for individual patients in the future. Here we describe fundamental aspects of electrical biosensing and review recent developments of the FET-based biotransistors.
2012
Preparation of α-mannoside hydrogel and electrical detection of saccharide-protein interactions using the smart gel-modified gate field effect transistor
Maeda, Y.; Matsumoto, A.; Miura, Y.; Miyahara, Y.
Nanoscale Res. Lett. 2012, 7, 1-8.
AbstractThe purpose of this study was to detect saccharide-protein interaction capitalizing on the gel-modified field effect transistor [FET]. A lectin-sensitive polymer gel that undergoes volume changes in response to the formation of molecular complex between ‘pendant’ carbohydrate and a ‘target’ lectin concanavalin A [Con A] was synthesized. It was revealed that direction and magnitude of the gel response (swelling or deswelling) could be readily designed depending on composition and network density of the gel. The Con A-sensitive polymer gel has shown the ability to transduce the detection of saccharide-protein interactions into electrical signals for FET.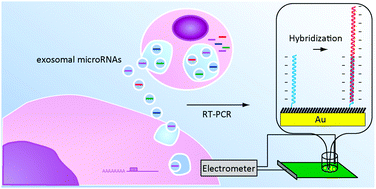 A label-free electrical detection of exosomal microRNAs using
microelectrode array
A label-free electrical detection of exosomal microRNAs using
microelectrode array
Goda, T.; Masuno, K.; Nishida, J.; Kosaka, N.; Ochiya, T.; Matsumoto, A.; Miyahara, Y.
Chem. Commun. 2012, 48(98), 11942-11944.
Abstract/Cover We report a method for detecting microRNAs encapsulated in exosomes
using a microelectrode array in semiconductor-based potentiometry after
RT-PCR. The inherent miniaturization of the electrical biosensor meets
requirements for massively parallel analysis of circulating microRNA as
a noninvasive biomarker.
We report a method for detecting microRNAs encapsulated in exosomes
using a microelectrode array in semiconductor-based potentiometry after
RT-PCR. The inherent miniaturization of the electrical biosensor meets
requirements for massively parallel analysis of circulating microRNA as
a noninvasive biomarker.
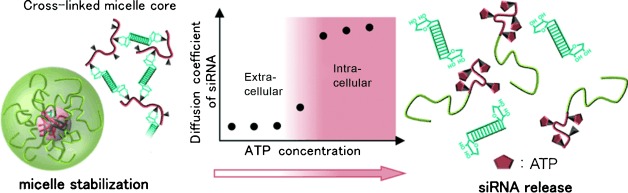 A Phenylboronate-Functionalized Polyion Complex Micelle for
ATP-Triggered Release of siRNA
A Phenylboronate-Functionalized Polyion Complex Micelle for
ATP-Triggered Release of siRNA
Naito, M.; Ishii, T.; Matsumoto, A.; Miyata, K.; Miyahara, Y.; Kataoka, K.
Angew. Chem.-Int. Edit. 2012, 51(43), 10751-10755.
Abstract/Cover PIC-ing a winner: siRNA encapsulated by a phenylboronate-functionalized polyion complex (PIC) micelle shows binding between the phenylboronate and the 3′ ribose of the siRNA (see scheme), stabilizing the complex under conditions equivalent to the extracellular environment. This complex is disrupted in response to addition of ATP, at a concentration comparable to that inside cells.
PIC-ing a winner: siRNA encapsulated by a phenylboronate-functionalized polyion complex (PIC) micelle shows binding between the phenylboronate and the 3′ ribose of the siRNA (see scheme), stabilizing the complex under conditions equivalent to the extracellular environment. This complex is disrupted in response to addition of ATP, at a concentration comparable to that inside cells.
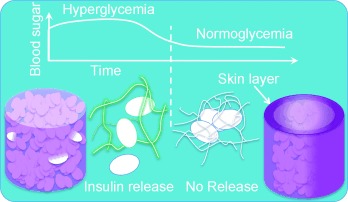 A Synthetic Approach Toward a Self-Regulated Insulin Delivery System
A Synthetic Approach Toward a Self-Regulated Insulin Delivery System
Matsumoto, A.; Ishii, T.; Nishida, J.; Matsumoto, H.; Kataoka, K.; Miyahara, Y.
Angew. Chem.-Int. Edit. 2012, 51(9), 2124-2128.
Abstract/Cover Protein-free: A hydrogel containing phenylboronate was optimized so as to undergo rapid glucose-dependent changes in the state of hydration under physiological aqueous conditions. A localized dehydration of the gel surface to form a “skin layer” enabled control of the release of insulin from the gel. This dehydration is induced by fluctuations in the glucose concentration in the range between normo- and hyperglycemia.
Protein-free: A hydrogel containing phenylboronate was optimized so as to undergo rapid glucose-dependent changes in the state of hydration under physiological aqueous conditions. A localized dehydration of the gel surface to form a “skin layer” enabled control of the release of insulin from the gel. This dehydration is induced by fluctuations in the glucose concentration in the range between normo- and hyperglycemia.
Reviews
自律型インスリン投与デバイスの開発状況と将来展望 (レビュー) (Current Development Status and Perspective of Self-Regulated Insulin Delivery Systems: A Review)
松元 亮; 宮原 裕二.
電気学会論文誌E (センサ・マイクロマシン部門誌) (IEEJ Transactions on Sensors and Micromachines) 2012, 132(12), 455-458.
AbstractDiabetes is not an infectious disease but its increasingly rapid and worldwide prevalence has been recognized as "pandemic". Despite the necessity for continuous and accurate glycemic control in the management of insulin dependent diabetes mellitus, the current palliative treatment relies almost solely on the patient-self injection of insulin, which not only impinges on quality of life of the patients but also fails to precisely control dose of insulin where the overdose must be strictly avoided otherwise causing serious hypoglycemia. Development of self-regulated insulin delivery systems is a long-standing challenge of materials science, for which exploitations of glucose oxidase and sugar-binding lectin are two prevalent strategies to install the function of glucose-sensitivity. These protein-based components, however, intolerant of long-term use and storage with their denaturing and cytotoxic natures, are hardly suitable for any implantable applications thus have not yet been in clinical usage to date. This review intends to provide an overview of the current efforts to develop a thoroughly synthetic alternative making use of a phenylboronic acid derivatized polyacrylamide gel.
Books
Intelligent Surfaces for Field-Effect Transistor-Based Nanobiosensing
Matsumoto, A.; Miyahara, Y.; Kataoka, K.
Intelligent Surfaces in Biotechnology: Scientific and Engineering Concepts, Enabling Technologies, and Translation to Bio‐Oriented Applications 2012, Chapter 4, 123-140 (John Wiley & Sons, Ltd).
AbstractSummary:- Introduction
- FET-Based Biosensors
- Intelligent Surfaces for Signal Transduction and Amplifi cation of bio-FETs
- New Targets of bio-FETs
- Future Perspective
- References
2011
Toward a Continuous Intravascular Glucose Monitoring System
Beier, B.; Musick, K.; Matsumoto, A.; Panitch, A.; Nauman, E.; Irazoqui, P.
Sensors 2011, 11(1), 409-424.
AbstractProof-of-concept studies that display the potential of using a glucose-sensitive hydrogel as a continuous glucose sensor are presented. The swelling ratio, porosity, and diffusivity of the hydrogel increased with glucose concentration. In glucose solutions of 50, 100, 200, and 300 mg/dL, the hydrogel swelling ratios were 4.9, 12.3, 15.9, and 21.7, respectively, and the swelling was reversible. The impedance across the hydrogel depended solely on the thickness and had an average increase of 47 Omega/mm. The hydrogels exposed to a hyperglycemic solution were more porous than the hydrogels exposed to a normal glycemic solution. The diffusivity of 390 Da MW fluorescein isothiocyanate in hydrogels exposed to normal and hyperglycemic solutions was examined using fluorescence recovery after photobleaching and was found to be 9.3 x 10(-14) and 41.4 x 10(-14) m(2)/s, respectively, compared to 6.2 x 10(-10) m(2)/s in glucose solution. There was no significant difference between the permeability of hydrogels in normal and hyperglycemic glucose solutions with averages being 5.26 x 10(-17) m(2) and 5.80 x 10(-17) m(2), respectively, which resembles 2-4% agarose gels. A prototype design is presented for continuous intravascular glucose monitoring by attaching a glucose sensor to an FDA-approved stent.
Reviews
バイオセンサの現状と今後の展望 (Trend and Perspective of Biosensors)
松元 亮; 宮原 裕二.
応用物理 (Applied Physics) 2011, 80(3), 205-210.
Abstract昨今,エレクトロニクスと医学・生物学の融合領域として著しい発展をみせるバイオセンサに関する研究の現状と将来展望について解説する.さまざまなナノ構造を利用して新たな生体検出の原理を創出する試みや,生体分子を標識なしに電気的に検出しうることで近年注目されているバイオトランジスタの取り組みについてその現状と将来の動向を展望する。ナノバイオセンシングの新展開 (New Trends in NanoBio-Sensing)
松元 亮; 宮原 裕二.
Drug Deliv. Syst. 2011, 26(1), 15-19.
Abstractエレクトロニクスと医学・生物学の融合領域として著しい発展をみせるバイオセンサに関する最新の研究動向を概説する. さまざまなナノ構造を利用し, 高感度に生体分子を検出する原理を創出する試みや, 生体分子を標識なしに電気的に検出しうることで近年注目されるバイオトランジスタの取り組みについて, 最新のアプローチを紹介し, 将来展望を述べる。
2010
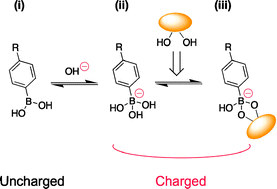 A totally synthetic glucose responsive gel operating in physiological
aqueous conditions
A totally synthetic glucose responsive gel operating in physiological
aqueous conditions
Matsumoto, A.; Yamamoto, K.; Yoshida, R.; Kataoka, K.; Aoyagi, T.; Miyahara, Y.
Chem. Commun. 2010, 46(13), 2203-2205.
Abstract/CoverHydrogel-based field effect transistor for lectin detection
Maeda, Y.; Matsumoto, A.; Miura, Y.; Miyahara, Y.
Eur. Cells Mater. 2010, 20(SUPPL.3), 167.
Bio-transistor for Label Free Living Cell Diagnosis
Matsumoto, A.; Sato, N.; Yoshida, R.; Kataoka, K.; Miyahara, Y.
Eur. Cells Mater. 2010, 20(SUPPL.3), 173.
Development of cell/transistor interface for real-time and noninvasive monitoring of potassium ion release based on apoptosis using biologically-coupled field effect transistor
Murakami, T.; Sakata, T.; Matsumoto, A.; Takai, M.; Ishihara, K.; Miyahara, Y.
Trans. Mat. Res. Soc. Jpn 2010, 35(2), 255-258.
AbstractWe report the real-time and noninvasive detection of K+ release through cell membrane using a biologically-coupled field effect transistor (bio-FET). To achieve this purpose, the cell/transistor interface, which can selectively detect K+ release through the ion-channel, was developed. The K+ release through the ion-channel caused by apoptosis (programmed cell death) could be electrically detected using the bio-FET with crown ether monolayer. Thus we have demonstrated the ability to analyze selectively the ion channel behavior using the bio-FET modified with the functional monolayer.Self-assembled molecular gate field effect transistor for label free sialic acid detection at cell membrane
Matsumoto, A.; Sato, N.; Cabral, H.; Kataoka, K.; Miyahara, Y.
Procedia Eng. 2010, 5, 926-929.
AbstractThis work describes a label free, potentiometric method to detect cell surface sialic acid (SA) using phenylboronic acid (PBA) compound integrated into the form of self-assembled monolayer (SAM) on a field effect transistor (FET) extended gold gate electrode. Due to predominant binding between undisassociated PBA and SA at pH 7.4, we found that carboxyl anions of SA were exclusively detectable among other glycan chain constituent monosaccharides, b as the change in threshold voltage (VT) of the PBA-modified FET. The technique was applied to analyses of altered SA expressions on rabbit erythrocyte as a models for diabetes. The comparative analyses revealed that the disease could be feasibly diagnosed simply by placing the cell suspensions onto the device without any labelling and enzymatic procedures.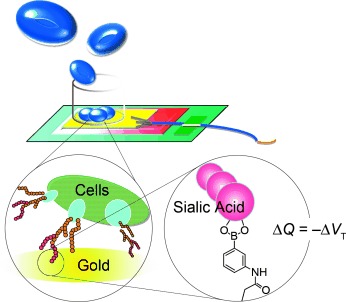 Assessment of Tumor Metastasis by the Direct Determination of
Cell-Membrane Sialic Acid Expression
Assessment of Tumor Metastasis by the Direct Determination of
Cell-Membrane Sialic Acid Expression
Matsumoto, A.; Cabral, H.; Sato, N.; Kataoka, K.; Miyahara, Y.
Angew. Chem.-Int. Edit. 2010, 49(32), 5494-5497.
Abstract Press releaseIn a world without labels…︁ A self-assembled phenylboronic acid monolayer on the surface of a gold electrode enabled the potentiometric detection of cell-membrane sialic acid (SA; see diagram) and differentiation of healthy pneumocytes from metastatic melanoma cells on the basis of altered SA expression. This approach to the diagnosis of tumoral malignancy or the degree of metastasis holds promise for label-free, noninvasive, real-time cytology.
Reviews
バイオトランジスタの開発 (Development of Biotransistors)
松元 亮; 宮原 裕二.
現代科学 (Modern Science) 2010, 6(471), 54-58.
Abstract生体分子が帯びている微弱な電荷を直接検出できるバイオトランジスターの開発と, 各種デバイスへの応用を紹介。
Books
バイオトランジスタによる生体分子認識の検出 (Detection of Biomolecule recognition using Biotransistors)
宮原 裕二; 坂田 利弥; 松元 亮; 片岡 知歩.
バイオチップ実用化ハンドブック (Handbook of Biochip Technology) 2010, Chapter 4, 518-528 (NTS Inc.).
薬物放出ゲル (Drug Release Gel)
松元 亮; 宮原 裕二.
食品・化粧品・医療分野へのゲルの利用 (Utilization of Gels in Food,Cosmetics and Medicine) 2010, Chapter 6, 240-245 (CMC Publishing Co., Ltd).
2009
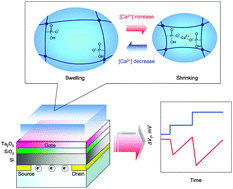 Electrical visualization of chemo-mechanical signal transduction using a
smart gel-modified gate field effect transistor
Electrical visualization of chemo-mechanical signal transduction using a
smart gel-modified gate field effect transistor
Matsumoto, A.; Endo, T.; Yoshida, R.; Miyahara, Y.
Chem. Commun. 2009, 5609-5611.
AbstractA field effect transistor (FET) whose gate surface has been modified with a stimuli-responsive “smart” polymer gel can visualize the kinetics of the chemo-mechanical signal transduction as a mode of its altered electrical characteristics without any labels.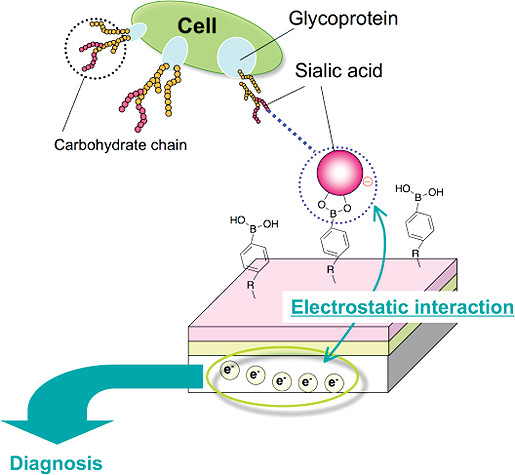 Noninvasive Sialic Acid Detection at Cell Membrane by Using
Phenylboronic Acid Modified Self-Assembled Monolayer Gold Electrode
Noninvasive Sialic Acid Detection at Cell Membrane by Using
Phenylboronic Acid Modified Self-Assembled Monolayer Gold Electrode
Matsumoto, A.; Sato, N.; Kataoka, K.; Miyahara, Y.
J. Am. Chem. Soc. 2009, 131(34), 12022-12023.
AbstractAlternations of sialic acid (SA) content on cell surface glycan chains have been implicated in numerous normal and pathological processes including developments, differentiations, and tumor metastasis. Overexpressions of SA have been implicated in the malignant and metastatic phenotypes for many different types of cancers, while decreased SA expression has also been identified in erythrocytes of diabetic mellitus. Techniques to conveniently monitor cell surface SA would therefore have great relevance to cytology. Preexisting methodologies to quantify SA, however, involve multiple enzymatic, dye-labeling, and lethal procedures, which are costly and time-consuming. Here we developed a potentiometric SA detection using a phenytboronic acid (PBA) compound integrated into the form of a self-assembled monolayer (SAM) onto a field effect transistor (FET) extended gold gate electrode. Due to selective binding between undisassociated PBA and SA at pH 7.4 among other glycan chain constituent monosaccharides, we found that carboxyl anions of SA were exclusively detectable as the change in threshold voltage (V-T) of the PBA-modified FET. The technique was applied to analyses of altered SA expressions on rabbit erythrocyte as a model for diabetes. Comparative SA expression analyses for each healthy and diseased model revealed that the disease could be feasibly diagnosed simply by placing the known-count cell suspensions onto the device without any labeling and enzymatic procedures. Such a technique may also provide a quantitative adjunct to histological evaluation of tumor malignancy and metastatic potential during intra- and postoperative diagnoses. Also advantageously, a technique herein described is all within a CMOS (Complementary Metal Oxide Semiconductor) compatible format thus promising for highly efficient and tow cost manufacturing with readiness of downsizing and integration by virtue of advanced semiconductor processing technologies.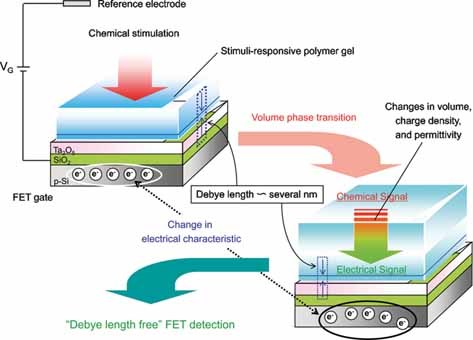 Chemical-to-Electrical-Signal Transduction Synchronized with Smart Gel
Volume Phase Transition
Chemical-to-Electrical-Signal Transduction Synchronized with Smart Gel
Volume Phase Transition
Matsumoto, A.; Sato, N.; Sakata, T.; Yoshida, R.; Kataoka, K.; Miyahara, Y.
Adv. Mater. 2009, 21(43), 4372-4378.
AbstractA stimulus-responsive polymer gel designed on a field-effect transistor gate undergoes a reversible volume phase transition in response to a specific biomolecule. An abrupt permittivity change at the gel/gate interface during the transition gives rise to a chemical to electrical signal conversion; the signal is thus detectable via a transistor without the limit of the Debye length.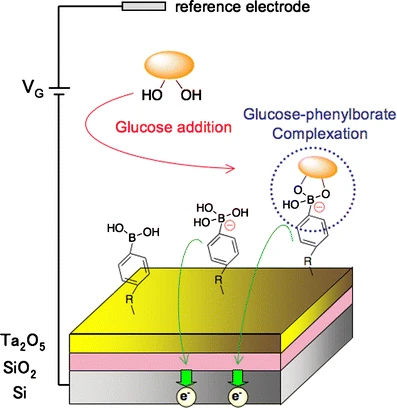 Glucose-sensitive field effect transistor using totally synthetic
compounds
Glucose-sensitive field effect transistor using totally synthetic
compounds
Matsumoto, A.; Sato, N.; Sakata, T.; Kataoka, K.; Miyahara, Y.
J. Solid State Electrochem. 2009, 13(1), 165-170.
AbstractA field effect transistor (FET)-based glucose sensor was fabricated. As a totally synthetic and thus stable glucose-sensing moiety, 3-acrylamidophenylboronic acid was chemically introduced onto the FET gate surface in the form of a thin copolymer gel layer. Excellent transistor characteristics were confirmed even after the surface modification, ensuring validity of the modification procedure herein developed. Glucose-induced changes in the FET’s electric characteristics were obtained in quantitative as well as reversible manners. It was also demonstrated that the prepared FET is able to continuously perceive the change in the glucose concentration in the milieu. The detected signals were attributed to the faction change of the gate-introduced phenyborate anions, also presumably involving other parameter changes such as permittivity and conductivity. The use of the fabricated FET could further be extended to the construction of stable, readily minutualizable, and label-free carbohydrate molecule-sensing systems.Label free carbohydrate detection by using phenylboronic acid gate-modified field effect transistor
Matsumoto, A.; Sato, N.; Miyahara, Y.
Curr. Appl. Phys. 2009, 9(4, 1), E214-E217.
AbstractThis paper describes a field effect transistor (FET) based sugar molecule sensor, using a totally synthetic and chemically stable sugar-detecting moiety phenylboronic acid (PBA). PBA was introduced to the FET sensing surfaces in two different ways. One is in the form of a thin copolymer gel layer directly attached to the FET gate insulator, the other method utilizes self-assembled monolayer (SAM) formed on an extended Au gate surface, a functional group of which was reacted with PBA. Reversible and prompt changes in the FET’s electric characteristics were demonstrated in response to the change in glucose and sialic acid (Neu5Ac) concentrations depending on the pH of the milieu. The developed FET detection scheme could be extended to the construction of stable, readily miniaturizable, and label-free carbohydrate molecule sensing systems due to the ability of PBA moieties to interact with these molecules. (C) 2009 Elsevier B.V. All rights reserved.
Books
ソフトマターを用いるバイオデバイス (Biodevices using semiconductor materials)
松元 亮; 宮原 裕二.
ソフトマター ~分子設計・キャラクタリゼーションから機能性材料まで~ (Soft matter - From molecular design and characterization to functional materials) 2009, Chapter 4.3, 222-232 (Maruzen Publishing Co. Ltd).
半導体技術を応用したバイオデバイス (Biodevices using semiconductor materials)
松元 亮; 宮原 裕二.
ナノ空間材料の創製と応用 (Nanospace Materials - Fundamentals and Applications) 2009, Chapter 4, 174-183 (Frontier Publishing Co. Ltd).
バイオトランジスタを用いた生体分子検出チップ (Bio-molecular detection chip using biotransistors)
松元 亮; 宮原 裕二.
ヘルスケアとバイオ医療のための先端デバイス機器 (Advanced Bio/Medical Devices and Equipments for Health Care) 2009, Chapter 1, 3-12 (CMC Publishing Co., Ltd).
2008
Silk Fibroin Solution Properties Related to Assembly and Structure
Matsumoto, A.; Lindsay, A.; Abedian, B.; Kaplan, D. L.
Macromol. Biosci. 2008, 8(11), 1006-1018.
AbstractThe impact of physiological factors on silk fibroin solution properties was studied. Specifically, the impact of fibroin concentration, protein purity, cation type and concentration, and pH on aqueous solution viscosity, shear behavior, and surface tension were assessed in the context of silk protein assembly. The results demonstrate that in vitro results could be correlated to in vivo processing events during silk spinning. Rheological properties with reference to the amphiphilic block structure of the protein are described, pH dependency of shear response was quantitatively correlated to the predicted pI values of the fibroin protein, and cooperativity among environmental factors such as pH and salts was identified. Stabilization of silk fibroin solution states by calcium was identified as a mode to control shear sensitivity of the fibroin solution. The cooperativities identified suggest tight control of fibroin aqueous solution rheological properties to gain a window of protection against premature crystallization of the fibroin during processing, assuring sate storage, transport, and finally successful fiber spinning.
Books
Microbial Genetic Analysis Based on Field Effect Transistors
Miyahara, Y.; Sakata, T.; Matsumoto, A.
Principles of Bacterial Detection: Biosensors, Recognition Receptors and Microsystems 2008, 313-339 (Springer).
AbstractIn this chapter, potentiometric detection methods for microbial DNA involved recognition events by use of genetic field effect devices will be described. Fundamental principles of field effect devices and the technical background with their ongoing applications in the field of bio-sensor technologies, termed bio-FET, will be first introduced. Then concept of genetic field effect transistor will be described with emphasis on their fabrication, characteristics, and recent applications to microbial Single Nucleotide Polymorphysms (SNPs) Analysis as well as DNA sequencing. By comparing to other conventional methods, technical significance and future perspective of the genetic field effect transistor will also be discussed in detail.
2007
Silk microspheres for encapsulation and controlled release
Wang, X.; Wenk, E.; Matsumoto, A.; Meinel, L.; Li, C.; Kaplan, D. L.
J. Control. Release 2007, 117(3), 360-370.
AbstractA method was developed to prepare silk fibroin microspheres using lipid vesicles as templates to efficiently load protein drugs in active form for controlled release. The lipid was subsequently removed by methanol or sodium chloride treatments, resulting in silk microspheres consisting of beta-sheet structure and about 2 mu m in diameter. NaCl treated microspheres had smoother surfaces compared to the methanol treatments based on SEM analysis, and both types of microspheres had a mixture of multilamellar and unilamellar structures. A model protein drug, horseradish peroxidase, was encapsulated in the microspheres. Freeze-thaw cycles during preparation led to higher loading of the peroxidase due to improved mixing between the silk and drug, while without this process the drug and silk remained in separate layers or domains in microspheres. This partitioning was determined with fluorescein-labeled silk and rhodamine-labeled dextran. Small molecules such as the enzyme substrate 3,31,5,5’-tetramethylbenzidine, Mw = 240 Da, and its oxidized product freely diffused through the MeOH- and NaCl-processed silk microspheres so that enzyme loading and activity could be determined. Enzyme activity was retained during processing and in the final microspheres. The enzyme release profile depended on the NaCl-process used in microsphere preparation. The physically cross-linked P-sheet structure of silk fibroin and the residual lipids in the microspheres played important roles in controlling enzyme release profiles. The silk microspheres have the potential for diverse applications where controlled protein release from biocompatible, mechanically tough, and slowly biodegradable carriers is desirable. (c) 2006 Elsevier B.V. All rights reserved.Silk fibroin microfluidic devices
Bettinger, C. J.; Cyr, K. M.; Matsumoto, A.; Langer, R.; Borenstein, J. T.; Kaplan, D. L.
Adv. Mater. 2007, 19(19), 2847-2850.
AbstractMicrofluidic devices were fabricated from silk ribroin using an aqueous molding process adapted from soft lithography. The process results in mechanically robust, patent devices with macroscopic fluidic connections. Hepatocytes were seeded in the device and perfused for up to five days during which they exhibited liver-specific functions equivalent to that observed for standard tissue culture substrates.
Reviews
電界効果を基本原理としたバイオセンシングデバイス (Biosensing device based on electric field effect as the basic principle)
坂田 利弥; 松元 亮; 宮原 裕二.
マテリアルインテグレーション (Materials integration) 2007, 20(9), 50-56.
遺伝子解析用バイオトランジスタ (Bio-transistor for Genetic Analysis)
坂田 利弥; 松元 亮; 宮原 裕二.
機能材料 ~ナノ・マイクロ技術を用いたバイオデバイス~ (Functional materials - Biodevices using nano/micro technology) 2007, 27(5), 25-34 (CMC Publishing Co., Ltd).
Abstract近年,ヒトゲノムの全塩基配列解読が完了しポストゲノム時代を迎え,創薬開発やテーラーメード医療をはじめ,さまざまな産業においてバイオチップの貢献が期待されている。これらのチップでは,生体分子の特異的反応の検出に蛍光,屈折率,質量,酸化・還元電流,化学発光などが用いられている。本稿では,これらの手法とは異なり,生体分子固有の電荷を電界効果により検出するバイオトランジスタについて紹介し,特にDNA分子認識反応を検出する遺伝子トランジスタについて筆者らの研究を中心に説明する。また,上記遺伝子トランジスタを,一塩基多型(SNPs)やDNAシーケンシング解析に応用する試みについても紹介する。
2006
Mechanisms of silk fibroin sol-gel transitions
Matsumoto, A.; Chen, J.; Collette, A. L.; Kim, U.-J.; Altman, G. H.; Cebe, P.; Kaplan, D. L.
J. Phys. Chem. B 2006, 110(43), 21630-21638.
AbstractSilk fibroin sol-gel transitions were studied by monitoring the process under various physicochemical conditions with optical spectroscopy at 550 nm. The secondary structural change of the fibroin from a disordered state in solution to a beta-sheet-rich conformation in the gel state was assessed by FTIR and CD over a range of fibroin concentrations, temperatures, and pH values. The structural changes were correlated to the degree of gelation based on changes in optical density at 550 nm. No detectable changes in the protein secondary structure (FTIR, CD) were found up to about 15% gelation (at 550 nm), indicating that these early stages of gelation are not accompanied by the formation of beta-sheets. Above 15%, the fraction of beta-sheet linearly increased with the degree of gelation. A pH dependency of gelation time was found with correlation to the predominant acidic side chains in the silk. Electrostatic interactions were related to the rate of gelation above neutral pH. The overall independencies of processing parameters including concentration, temperature, and pH on gel formation and protein structure can be related to primary sequence-specific features in the molecular organization of the fibroin protein. These findings clarify aspects of the self-assembly of this unique family of proteins as a route to gain control of material properties, as well as for new insight into the design of synthetic silk-biomimetic polymers with predictable solution and assembly properties.Fibrous proteins and tissue engineering
Wang, X.; Kim, H. J.; Wong, C.; Vepari, C.; Matsumoto, A.; Kaplan, D. L.
Mater. Today 2006, 9(12), 44-53.
AbstractFibrous proteins are finding broad impact in biomaterial systems for a range of cell and tissue studies. This impact derives from an improved insight into fundamental structure-function relationships, as well as the unique material properties attained with these protein polymers. Recent advances in the use of these protein systems in a variety of biomaterial and tissue engineering applications are reviewed, with a focus on approaches to control the structure, chemistry, and morphology of the biomaterials formed and enable cell and tissue outcomes to be directed in vitro and in vivo.
Books
Silk
Matsumoto, A.; Kim, H. J.; Tsai, I.; Wang, X.; Cebe, P.; Kaplan, D. L.
Handbook of Fiber Chemistry Third Edition 2006, 383-404 (CRC Press).
AbstractSilk is an externally spun fibrous protein secretion formed into fibers, usually resulting in material structures such as cocoons or webs [1-4]. Silks are essentially pure proteins, with only in some Of all the natural silks represent the only ones that are spun. Silk fibers from silkworms have been used in textiles for nearly 5000 years. The primary reasons for this longtime use have been the unique luster, tactile properties, durability, and dyability of silks. Silk fibers are remarkable materials displaying unusual mechanical properties: strong, extensible, and mechanically compressible. Silks also display interesting thermal and electromagnetic responses, particularly in the UV range for insect entrapment and form crystalline phases related to processing. Silk fibers were used in optical instruments as late as the mid-1900s because of their fine and uniform diameter and high strength and stability over a range of temperatures and humidity. Naturalist reports suggest that some spider silks were used in the South Pacific for gill nets, dip nets, and fishing-a testimony to the remarkable mechanical properties and durability of this family of protein polymers. Silks have historically been used in medicine as sutures over the past 100 years and are currently used today in this mode along with a variety of consumer product applications. Commercially, silkworm cocoons are mass produced in a process termed ‘‘sericulture’’. The cocoons are extracted in hot soapy water to remove the sericin gluelike protein. The remaining fibroin or structural silk is reeled onto spools, yielding approximately 300-1200 m of usable thread per cocoon. These threads can be dyed or modified for textile applications. The annual world production of raw silk is about 60,000 tons, with China producing half of the world supply followed by India, Korea, and Japan. Silks represent one member of a larger class of fibrous proteins in nature, which include keratins, collagens, elastins, and others [5]. These types of proteins can be considered nature’s equivalent of synthetic block copolymers. Aside from their direct use in materials applications, fibrous proteins provide experimentally accessible model systems with simpler and well-controlled genetic template-based protein synthesis. The highly repetitive structure allows key features of the primary sequences of these proteins to be captured in shorter consensus sequences at the corresponding genetic level. Short synthetic genetic variants can then be combined to generate larger genes and thus proteins that represent mimics of the native protein. This technique is useful in simplifying the complex behavior of these proteins to an intelligible level, while retaining their biological relevance and materials function. These shorter genetic variants, when polymerized (multimerized) into longer genes, can be used to explore protein sequence and size relationships.絹タンパク質のナノスケールでの自己組織化と構造制御 (Nanoscale features of silkworm cocoon and spider orb web silk proteins)
松元 亮; Kaplan, D. L.
’ファイバー’スーパーバイオミメティックス~近未来の新技術創成~ (’Fiber’ Superbiomimetics - The Creative Technologies in Coming Future) 2006, Part 5, Chapter 1.4, 790-798 (NTS Inc.).
2005
Processing windows for forming silk fibroin biomaterials into a 3D porous matrix
Kim, H. J.; Kim, H. S.; Matsumoto, A.; Chin, I. J.; Jin, H. J.; Kaplan, D. L.
Aust. J. Chem. 2005, 58(10), 716-720.
AbstractIn the present study we clarify phase diagrams related to silk fibroin processing into three-dimensional porous structures useful for biomaterials and for scaffolds in tissue engineering. All-aqueous and organic solvent (hexafluoroisopropanol) modes of processing are compared relative to solution concentration of silk protein polymer and size of porogen (NaCl particles). The results clarify the range of conditions under which these biomaterial matrices can be formed, with a broader range of pore sizes and smoother surface morphology generated from the organic solvent process. These structures are directly applicable to fundamental studies of protein-based biomaterial assembly as well as cell interactions and tissue formation with these systems.Biomaterial coatings by stepwise deposition of silk fibroin
Wang, X. Y.; Kim, H. J.; Xu, P.; Matsumoto, A.; Kaplan, D. L.
Langmuir 2005, 21(24), 11335-11341.
AbstractA completely aqueous, stepwise deposition process with Bombyx mori silk fibroin for the assembly of nanoscale thin film coatings is reported the first time. The focus of this work was to develop an understanding of the control of this deposition process and to characterize the films formed from a physicochemical perspective. The deposition process was monitored by UV spectrophotometry and research quartz crystal microbalance. Both absorbance and film thickness correlated linearly with the number of silk fibroin layers deposited, analogous to multilayered materials fabricated from conventional polyelectrolytes. The polymer adsorption process was stable and reproducible, with control of a single layer thickness ranging from a few to tens of nanometers, determined by the concentrations of silk fibroin, salt concentration in the dipping solution, and method of rinsing. The driving force for the assembly of silk fibroin onto the substrate was primarily hydrophobic interactions, while some electrostatic interactions were also involved. The difference with this approach from traditional polyelectrolyte layer-by-layer techniques is that an intervening drying step is used to control the structure and stability of the self-assembled silk fibroin. The assembled films were stable under physiological conditions and supported human bone marrow stem cell adhesion, growth, and differentiation. This approach offers new options to engineer biomaterial coatings as well as bulk materials with control of both interfacial properties conducive to specific cellular or tissue responses and the potential to entrap and deliver labile molecules or other components due to the all-aqueous process described.
2004
Swelling and shrinking kinetics of totally synthetic, glucose-responsive polymer gel bearing phenylborate derivative as a glucose-sensing moiety
Matsumoto, A.; Kurata, T.; Shiino, D.; Kataoka, K.
Macromolecules 2004, 37(4), 1502-1510.
AbstractA glucose-responsive polymer gel bead with a diameter in the range of several hundred micrometers, bearing a phenylborate derivative (3-acrylamidophenylboronic acid) as a glucose-sensing moiety, was successfully prepared by inverse phase suspension polymerization. The kinetics of the glucose responsive swelling and shrinking process of the gel bead was studied by monitoring changes in the size and shape of the gel under a microscope. The equilibrium swelling volumes of the gel determined under various temperatures and glucose concentrations revealed the presence of critical temperatures and glucose concentrations to induce discontinuous volume phase transitions of the gel. The glucose-induced swelling process was accompanied by the appearance of a marked swelling boundary (swelling front) intervening between the core collapsed phase and the outer swollen layer of the gel, and the swelling curve as a function of the square root of the time exhibited a sigmoidal (nonlinear) feature, both indicating that the process is essentially rate determined by the relaxation process of the polymer chains due to the hydration. The swelling rate was significantly affected by the bead size and the terminal glucose concentration. An accelerated disappearance of the swelling front was observed immediately before the gel reaches the equilibrium swelling, which was highlighted by an increased bead size, implying that increasing elastic and osmotic pressures generating from the swollen layer affect the swelling kinetics. The glucose-induced shrinking process involved the formation of a skin layer on the gel surface followed by a radical structural change. The observed, appreciably long-termed preservation of a quasi-swollen state on a sub-millimeter scale gel bead with a distinctive skin layer may propose the potential applicability of the gel to chemical valve systems discretely switching for solute release.Glucose-responsive polymer gel bearing phenylborate derivative as a glucose-sensing moiety operating at the physiological pH
Matsumoto, A.; Yoshida, R.; Kataoka, K.
Biomacromolecules 2004, 5(3), 1038-1045.
AbstractThe work attempts to prepare a totally synthetic, glucose-responsive polymer gel bearing a phenylborate derivative as a sensor moiety to glucose, for future use as a self-regulated insulin delivery system. The molecular strategies to enable the system to be operated under physiological conditions (pH 7.4, 37 degreesC) are presented that involve the use of a novel pherylborate derivative {4-(1,6-dioxo-2,5-diaza-7-oxamyl) phenylboronic acid: DDOPBA} possessing an appreciably low pK(a) (similar to7.8), the adoption of poly(Nisopropylmethacrylamide) (PNIPMAAm) for the main chain, which itself undergoes a sharp thermo-induced phase transition at its LCST around 40 degreesC, as well as the introduction of a carboxyl group of methacrylic acid as the third comonomer. Glucose-responsive behaviors of the obtained gels were evaluated based on the changes in the equilibrium swelling degree determined in the presence and the absence of glucose, for various pH and temperature conditions. As a consequence of the combined molecular effects, a sufficient sensitivity of the system was accomplished at physiological pH and in the temperature range close to the physiological condition such as 30 degreesC. Furthermore, the glucose-induced continuous volume changes of the gels were demonstrated under those conditions, which occurred in a remarkably concentration-dependent manner. In these experiments, the critical glucose concentrations to induce the gels’ responses in the range of normoglycemic sugar level were observed. These observations may provide us with an excellent prospect for the use of the gel as a self-regulated, insulin-delivery system discretely switching the release at the normoglycemia.
2003
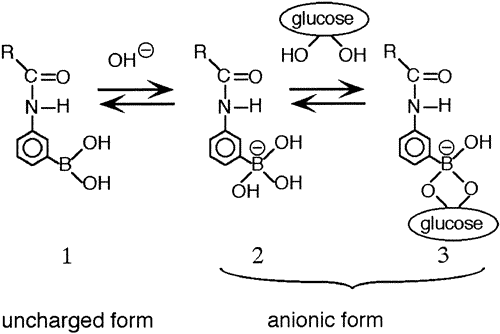 Glucose-responsive polymer bearing a novel phenylborate derivative as a
glucose-sensing moiety operating at physiological pH conditions
Glucose-responsive polymer bearing a novel phenylborate derivative as a
glucose-sensing moiety operating at physiological pH conditions
Matsumoto, A.; Ikeda, S.; Harada, A.; Kataoka, K.
Biomacromolecules 2003, 4(5), 1410-1416.
AbstractThis study is devoted to the development of novel glucose-responsive polymers that operate under physiological conditions (pH 7.4, 37 degreesC), aiming for future use in a self-regulated insulin delivery system to treat diabetes mellitus. The approach involves the use of a newly synthesized phenylborate derivative {4-(1,6-dioxo-2,5-diaza-7-oxamyl) phenylboronic acid, DDOPBA} possessing an appreciably low pK(a) (similar to7.8) as a glucose-sensing moiety, as well as the adoption of poly(N-isopropylmethacrylamide), PNIPMAAm, as the main chain that exhibits critical solution behavior in the range close to physiological temperature. Glucose-and pH-dependent changes in the critical solution behavior of the resultant copolymers were investigated at varying temperatures, revealing definite glucose sensitivities near the physiological conditions. Furthermore, DDOPBA moieties in the copolymers maintained constant apparent pK(a) values even when the temperature approaches the critical solution points of the main chain, indicating that spacing of the phenylborate moiety from the polymer backbone is a feasible way to minimize the microenvironment effect caused by a temperature-induced change in the hydration state of the polymer strands.


